| Original Article, Biomed Biopharm Res., 2023; 20(1):93-105 doi: 10.19277/bbr.20.1.315; PDF version [+]; Portuguese html [PT] |
Addressing the digital divide: who is being left behind in the evaluation of e-Health interventions to improve medication adherence?
João Gregório 1 ![]() ✉️, Tiago Maricoto 2
✉️, Tiago Maricoto 2 ![]() , Paulo A.S. Moreira 3
, Paulo A.S. Moreira 3 ![]() , Fátima Roque 4
, Fátima Roque 4 ![]() , Jaime Correia-de-Sousa 5
, Jaime Correia-de-Sousa 5 ![]() , Maria Teresa Herdeiro 6
, Maria Teresa Herdeiro 6![]() , Isabel F Almeida 7
, Isabel F Almeida 7 ![]() , Ioanna Tsiligianni 8
, Ioanna Tsiligianni 8 ![]() , Tamas Agh 9
, Tamas Agh 9 ![]() & Cristina Jácome 10
& Cristina Jácome 10 ![]()
1 CBIOS – Universidade Lusófona’s Research Center for Biosciences & Health Technologies, Lisboa, Portugal
2 Aveiro Healthcare Centre, Aradas Family Health Unit, Aveiro, Portugal; and Faculty of Health Sciences, University of Beira Interior, Covilhã, Portugal
3 Instituto de Psicologia e Ciências da Educação, Universidade Lusíada – Norte, Porto, Portugal; and Centro de Investigação em Psicologia para o Desenvolvimento(CIPD), Porto, Portugal
4 Research Unit for Inland Development, Polytechnic of Guarda (UDI-IPG), Guarda, Portugal
5 Life and Health Sciences Research Institute. School of Medicine. University of Minho. Braga, Portugal; and ICVS/3B’s - PT Government Associate Laboratory, Braga,Portugal
6 Institute of Biomedicine (iBiMED) and Department of Medical Sciences, University of Aveiro, Aveiro, Portugal
7 UCIBIO, REQUIMTE, Med Tech, Laboratory of Pharmaceutical Technology, Department of Drug Sciences, Faculty of Pharmacy, University of Porto, Porto, Portugal; andAssociate Laboratory i4HB - In-stitute for Health and Bioeconomy, Faculty of Pharmacy, University of Porto, Porto, Portugal
8 Department of Social Medicine, Faculty of Medicine, University of Crete, Crete, Greece
9 Syreon Research Institute, Budapest, Hungary; and Center for Health Technology Assessment and Pharmacoeconomic Research, University of Pécs, Pécs, Hungary
10 CINTESIS@RISE, MEDCIDS, Faculty of Medicine of the University of Porto, Porto, Portugal
Abstract
The implementation of e-Health initiatives aimed at improving medication adherence has shown limited success. This may be attributed to inadequate participant selection in evaluation studies. The objective of this evidence-based commentary is to examine the inclusion criteria of participants in e-Health interventions for medication adherence. In May 2021, we identified two systematic reviews, encompassing a total of 44 randomized controlled trials (RCTs) evaluating e-Health interventions for medication adherence. These interventions were aimed to diabetes (n=10), immunosuppressive therapy (n=6), and cardiovascular diseases (n=29). Participants in interventions utilizing digital apps had a lower mean age compared to those using telephones (53.9 ± 7.5 years vs. 63.4 ± 6.3 years). Most papers (91.0%) did not report participants' level of digital literacy, and educational background was only mentioned in 57.8% of the studies. Exclusion criteria associated with age, education, health conditions, were identified in the assessment of e-Health interventions, while digital literacy was seldom assessed. It is recommended incorporating the assessment and reporting of digital literacy into participant selection procedures in RCTs to address the digital divide and obtain more accurate information regarding the efficacy of e-Health interventions.
Keywords: Medication adherence, e-Health evaluation, digital divide, health inequities, digital health
To Cite: Gregório, J. et al. (2023) Addressing the digital divide: who is being left behind in the evaluation of e-Health interventions to improve medication adherence? Biomedical and Biopharmaceutical Research, 20(1), 93-105.
Correspondence to:
Received 18/04/2023; Accepted 12/07/2023
Introduction
With the expansion of life expectancy and better health care, an increase in the incidence of chronic diseases and multi-morbidity has been documented worldwide (1). Addressing multi-morbidity requires a multidisciplinary response, often involving lifestyle modifications combined with lifelong medication use and its associated complications (2). Among the major challenges faced by healthcare professionals, managing medication non-adherence stands out, as approximately 50% of patients with chronic diseases do not fully adhere to their therapeutic regimen (3). In Europe, non-adherence contributes to nearly 200,000 premature deaths and costs 125€ billion per year in avoidable hospitalizations, emergency care, and outpatient visits (4). In the United States, it is estimated that medication non-adherence causes approximately 125,000 deaths, accounts for 10% of hospitalizations, and results in healthcare system expenditures ranging from $100 billion to $289 billion annually (5). Due to this huge impact, medication adherence has been identified as a key priority in health policies and research agendas (6).
The term 'e-Health' encompasses the broad use of information and communication technology to enhance healthcare at local, regional, and global levels (7). Mobile health (m-Health) and wearable devices (such as smartwatches) are examples of e-Health technologies employed in medication adherence research, offering feasible integration into patients' daily lives (8,9). These e-Health interventions have the potential to empower patients in identifying and managing unintentional non-adherence and facilitate communication with healthcare professionals for shared decision-making (8,10).
However, the current evidence regarding the use of e-Health interventions to tackle medication non-adherence is diverse (11). Recent systematic reviews showed that the available evidence of the impact of e-Health solutions on medication non-adherence remains inconclusive (8,11–14). A number of factors might contribute to this situation, for example, the fact that most e-Health interventions propose one-size-fits-all solutions for distinct nonadherence patterns (erratic; unwitting; intelligent) (3). Another potential contributing factor is the inclusion of specific patient groups in the evaluation of e-Health interventions targeting medication adherence, which may not accurately represent the diverse spectrum of real-world patients. This concern has also been raised in other areas of e-Health research (15,16). Individuals with low health literacy are less prone to use e-Health tools and generally present worse health outcomes (17). Skills for using e-Health technologies depend on several factors, and some more vulnerable groups have been identified, such as those with lower literacy and elderly citizens (18).
The recognition that there is much to improve in this field, mainly due to the expected exponential growth of innovative solutions in the coming years, has led a group of researchers and professionals interested in all aspects of medication adherence to form a COST action (European Network to Advance Best practices and technology on medication adherencE -ENABLE, COST Action 19132). One of the aims of this action is to improve the knowledge on the application of novel technologies for medication adherence, their clinical applications, and policy measures supporting their implementation across Europe (19). Thus, the main goal of this paper is to contribute to the debate by providing an evidence-based commentary that examines the characteristics of participants enrolled in e-Health interventions focused on medication adherence and discuss the factors that can impact the evaluation of these interventions. By doing so, we aim to highlight how these factors can worsen inequities in e-Health for medication adherence. By doing so, we aim to contribute to the improvement of future research endeavors in this area.
Materials and Methods
To achieve the main aim of this paper, and considering the nature of this commentary, the authors searched for recent systematic reviews of randomized control trials concerning e-Health and medication adherence published in the Journal of Medical Internet Research (JMIR), a leading journal in the field of digital medicine and health. Determining this strategy was the research team’s perception of JMIR’s importance. In fact, the impact factor of this journal is 7,077 (source: JCR, 2021), being in the first quartile of the "Health Care Sciences and Services" category, and the first that also belongs to the "Medical Informatics" category. In this category, it is the most cited journal.
Search strategy
The search was performed in May 2021, and systematic literature reviews were selected if: published after 2020; included only randomized controlled trials; and described e-Health interventions in accordance with the e-Health definition of Eysenbach, that is “health services and information delivered or enhanced through the Internet and related technologies”(7). Systematic reviews were excluded if they reported papers that were population-specific.
Data analysis
After selecting the relevant systematic literature reviews, the individual studies supporting those reviews were collected. To ensure a comprehensive extraction of data from each article, we utilized a purpose-built form. This form included the main author’s last name and year of publication, country, health condition, interventions, eligibility criteria, sample size, and participants characteristics (age, gender, education, digital literacy, occupation, income, civil status, race). A team of seven reviewers (JG, TM, PM, JCS, MTH, FR, CJ) was responsible for extracting data from the selected articles, while two reviewers (JG and CJ) harmonized the extracted data to ensure consistency. Any discrepancies or disagreements among the reviewers regarding the collected data were discussed and resolved in monthly team meetings involving all authors.
Results
Collected papers
Two systematic reviews fulfilled the eligibility criteria: one with 21 papers and the other with 24 (13,14). These two reviews collectively analyzed a total of 45 papers, corresponding to 44 unique studies (20–63). All studies were randomized controlled trials and published between 2003 and 2020 (Supplementary material - Table S1). The majority of the studies (n=21) were conducted in the United States, followed by Western European countries (n=14). These studies addressed mostly e-Health interventions for patients with hypertension (n=12), diabetes (n=10) and with other cardiac conditions (n=8).
Participants’ characteristics – inclusion and exclusion criteria
Original studies used sociodemographic-, clinical-, and literacy-related eligibility criteria to select participants. In total, studies included 47,501 participants (samples between 37 and 21,752), with a mean age of 60±10 years. Inclusion of both men and women (n=42) with 18 years old or over (n=25) were the most common criteria. The distribution of male and female participants was relatively balanced, with females accounting for 47% and males for 53% of the total. Nine studies used other minimum age cut-offs (i.e., 7, 21, 40, 50, 60, and 65 years of age) and three studies even defined maximum cut-offs (i.e., ≥75, and ≥80 years of age). Apart from age, having specific medical conditions (n=38) or a specific medicine/groups of medicines prescribed (n=28) were the most frequent inclusion criteria. Other inclusion criteria were sub-optimum medication adherence (n=6), being followed up in a certain healthcare setting (n=8), and being able to self-manage medication at home (n=6).
With regard to literacy, criteria such as fluency in a specific language (n=12) and proficiency in using or accessing a smartphone (n=13), internet (n=8) or text messages (n=7) were used. Only 18 studies reported the educational levels of participants, and although various classifications were employed, the majority of participants had attained at least a higher education or completed more than 12 years of schooling. Fourteen studies provided information on ethnicity, including 12 from the USA, one from South America, and one from Germany. Across these studies, white participants were generally overrepresented, comprising 31% to 95% of the enrolled individuals. Marital status was reported in only six studies, with married participants constituting approximately half of the recruited individuals (ranging from 59% to 66%), except for one study, which reported a lower frequency (35%).
Exclusion criteria were mostly related to perceived difficulties with the technologies, either from cognitive and mental impairment (e.g., dementia; depression) (n=10); physical inability or other biological impairment (e.g., visual impairment) (n=6); specific medical treatments (n=6); living in an institution (n=6); participation in other studies (n=3) and low literacy (e.g., language and digital barriers, no other contact available) (n=3).
e-Health interventions
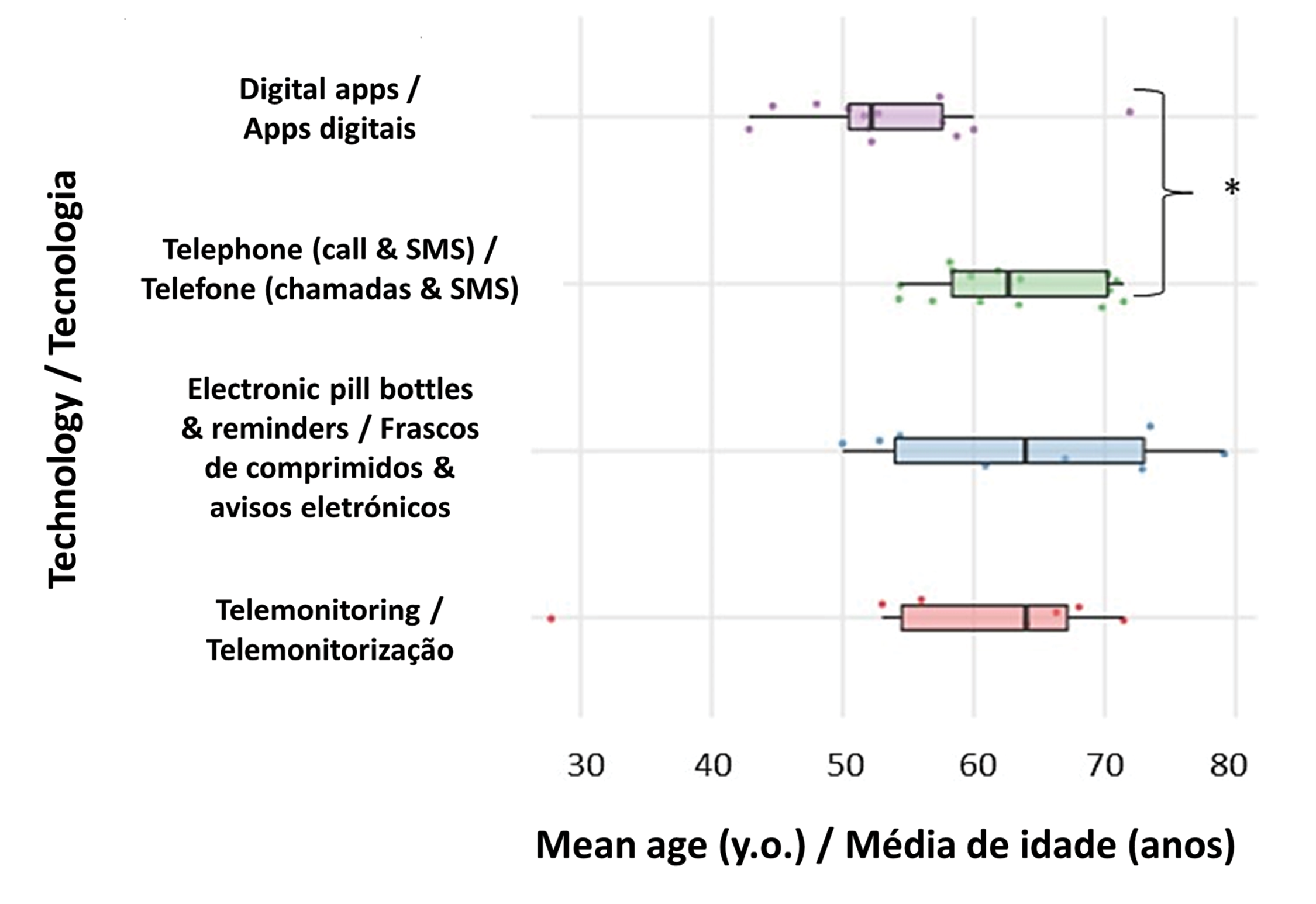
Regarding the types of e-Health interventions, a total of 21 studies employed telephone-based interventions to enhance medication adherence. These interventions consisted of stand-alone telephone calls (n=11), text messages (n=3), or a combination of telephone communication with pillboxes, reminders, interactive voice recognition, or tele-monitoring systems (n=7). Additionally, interventions utilizing stand-alone pill bottles/dispensers (n=5) and digital apps (n=8) were also common. Other interventions included the use of web-based tools (n=3), educational videos/books (n=2), video calls or webinars (n=2), and electronic reminders (n=1). It is worth noting that participants enrolled in studies using digital apps (including web-based tools, webinars, educational videos, or workbooks) or telemonitoring systems tended to be younger compared to those involved in studies utilizing "legacy" technologies such as the telephone (Figure 1). Notably, the majority of studies (91.0%) did not provide a characterization of participants' digital literacy.
|
Figure 1 - Participants’ mean age according to the type of intervention (* - ANOVA with the Tukey post hoc test; p-value=0.041). |
 |
Discussion
To our knowledge, this is the first study attempting to explore the selection criteria in the assessment of e-Health interventions directly concerning medication adherence.
Older and less educated patients may have been underrepresented in some of the analyzed studies, particularly in those with digital apps. The higher average age of participants in studies utilizing "legacy" technologies like the telephone highlights the continued reliance on 19th-century technology in the digital era to support e-Health services. It is widely recognized that age and education are factors associated with e-Health utilization (64,65). However, if these technologies are intended to contribute to improving medication adherence, they must be accessible across diverse age groups and literacy levels. Our analysis reveals a consistent exclusion of older adults, individuals with low literacy, and those with mental health issues from the evaluation of these technologies. This serves as evidence that the digital divide, in terms of age, literacy, and mental health, continues to persist (15,66).
The issue of the digital divide becomes even more significant when considering medication adherence, as older adults often have more complex medication regimens, which can affect their ability to utilize e-Health solutions effectively (67). Consequently, a more customized approach is necessary (68). It is therefore crucial to assess how these solutions perform specifically in this population, a task that has yet to be comprehensively addressed. This challenge is closely tied to the development of technological solutions. Typically, developers design applications in controlled laboratory environments and then anticipate their successful implementation in real-world scenarios. However, evaluating the efficacy of these innovations requires selecting patients who possess the necessary skills to utilize them. Many times, this is not possible since the patients to whom these innovations were addressed do not have the necessary skills. Older patients are seldom involved in the development of technological or digital solutions due to their complexity, hindering their application in real life (69). To overcome this barrier, resorting to participatory design methodologies and implementation research frameworks may be possible solutions to better tailor future digital solutions in the context of healthcare (70,71).
None of the studies examined in our analysis employed a standardized approach to assessing participants' confidence in using the technology, either for screening purposes or to characterize them. In our analysis, we found that the few studies that included digital literacy as eligibility criteria used broad definitions, such as access/use of a technology, but did not attempt to characterize participants’ capability of interacting with the proposed technology. Future research should use validated questionnaires to assess digital literacy, such as the e-Health Literacy Scale, the Media and Technology Usage and Attitudes Scale (72), among others. This could facilitate comparison among studies and better clarify how the inequities rising from the digital divide were approached. To effectively bridge this digital divide and minimize its impact on health equity, e-Health researchers must prioritize the integration of solutions aimed at enhancing digital literacy within their final interventions (73).
Ethnicity can be a determinant for eHealth access due to a combination of social, cultural, and structural factors, such as lower income levels, language barriers, or lack of diversity and cultural representation in the development of eHealth platforms, which can exacerbate disparities in health outcomes (74). In the studies analyzed, only those performed in the USA systematically collected data on the ethnicities of the participants. It is clear that most participants in all studies are white or of Caucasian ethnicity. None of the studies presented results on the effect of ethnicity on the results of the interventions. Moreover, up to 80% of the studies (35 out of 44) were performed in high-income countries (USA and Western European countries). This highlights the scarcity of research in low and middle-income countries, where the use of e-Health technologies, with the recognized potential to overcome the difficulties of dispersed geographic locations and rurality (66,75), may be more compromised (64,65). To some extent, this unmet need may delay the implementation of effective interventions for medication adherence.
Most evidence on the use of information technologies available is still sustained in observational studies or inadequately dimensioned experimental studies. This limitation significantly hampers the widespread implementation of e-Health applications within various health systems, as policymakers often require high-quality evidence to make informed decisions regarding the adoption of innovative solutions. Despite the mounting evidence highlighting the existence of the digital divide, it is evident that a concerted effort is necessary to expand our understanding of how interventions can be adapted to culturally diverse and economically disadvantaged settings. To achieve this, a shift towards an implementation research framework, as opposed to the traditional pure experimental approach, is called for. This approach will facilitate the exploration of contextual factors and enable the development of strategies that address the unique challenges posed by different populations and settings, thereby promoting more effective and equitable implementation of e-Health interventions (76). Another strategy that could provide more useful insights would be to perform realist reviews and synthesis of the literature, aiming to develop theories that can elucidate on when, why and how a certain program works (77).
Conducting a commentary based on individual studies extracted from only two recent systematic reviews on the topic may be seen as a significant limitation of the methodology we have used to sustain this work. Likewise, limiting our search to a single scientific journal (JMIR) can be seen as a limitation, but as previously mentioned, this is a leading journal in the field of digital medicine and it is not specific to any health condition, constituting an obvious choice for the authors. Nevertheless, we need to stress that our aim was to gather examples of the research produced on the topic and raise awareness about the inequities being introduced by the experimental assessment of e-Health interventions for medication adherence, in an attempt to contribute for improving future research efforts.
Conclusions
Inequities due to age, education, ethnicity, and digital literacy have been present in e-Health interventions. This digital divide still persists and little efforts have been made in recent years to tackle it. This hinders the assessment of the true impact of e-Health interventions concerning medication adherence in patients with multimorbidities and polymedication. It is, therefore, critical to foster equity in future research in this field, ensuring that technologies to improve adherence are evaluated inclusively by real-world patients, families, and caregivers.
Authors' Contributions Statement
JG and CJ conceived, coordinated the study and wrote the first draft of the manuscript. JG,TM, PM, FR, JCS, MTH, and CJ extracted the information from the original reviews, and contributed to the writing of all subsequent drafts of the manuscript. IT, IA and TA have contributed to the last draft of the manuscript. JG edited the final version. All authors read and provided significant inputs into the drafts of the manuscript, agreed to be accountable for all aspects of the work and approved the final draft of the manuscript for publication.
Funding
This research is based upon work from COST Action CA19132 “ENABLE,” funded by COST (European Cooperation in Science and Technology). The funder had no role in the study design, data collection, analysis and interpretation, or preparation of the manuscript. João Gregório is funded by Foundation for Science and Technology (FCT) Scientific Employment Stimulus contract with the reference number CEEC/CBIOS/EPH/2018.
Conflict of Interests
The authors declare there are no financial and/or personal relationships that could present a potential conflict of interests.
References
1. Nguyen, H., Manolova, G., Daskalopoulou, C., Vitoratou, S., Prince, M., & Prina, A. M. (2019). Prevalence of multimorbidity in community settings: A systematic review and meta-analysis of observational studies. Journal of Comorbidity, 9, 2235042X1987093. https://doi.org/10.1177/2235042X19870934
2. Sánchez-Fidalgo, S., Guzmán-Ramos, M. I., Galván-Banqueri, M., Bernabeu-Wittel, M., & Santos-Ramos, B. (2017). Prevalence of drug interactions in elderly patients with multimorbidity in primary care. International Journal of Clinical Pharmacy, 39(2), 343–353. https://doi.org/10.1007/s11096-017-0439-1
3. World Health Organization. (2003). Adherence to long-term therapies: Evidence for action / edited by Eduardo Sabaté
4. Khan, R., & Socha-Dietrich, K. (2018). Investing in medication adherence improves health outcomes and health system efficiency: Adherence to medicines for diabetes, hypertension, and hyperlipidaemia | OECD Health Working Papers | OECD iLibrary. https://www.oecd-ilibrary.org/social-issues-migration-health/investing-in-medication-adherence-improves-health-outcomes-and-health-system-efficiency_8178962c-en
5. Viswanathan, M., Golin, C. E., Jones, C. D., Ashok, M., Blalock, S. J., Wines, R. C. M., Coker-Schwimmer, E. J. L., Rosen, D. L., Sista, P., & Lohr, K. N. (2012). Interventions to Improve Adherence to Self-administered Medications for Chronic Diseases in the United States. Annals of Internal Medicine, 157(11), 785. https://doi.org/10.7326/0003-4819-157-11-201212040-00538
6. van Boven, J. F. M., Lavorini, F., Dekhuijzen, P. N. R., Blasi, F., Price, D. B., & Viegi, G. (2017). Urging Europe to put non-adherence to inhaled respiratory medication higher on the policy agenda: A report from the First European Congress on Adherence to Therapy. European Respiratory Journal, 49(5), 1700076. https://doi.org/10.1183/13993003.00076-2017
7. Eysenbach, G. (2001). What is e-Health? Journal of Medical Internet Research, 3(2), e20. https://doi.org/10.2196/jmir.3.2.e20
8. Tabi, K., Randhawa, A. S., Choi, F., Mithani, Z., Albers, F., Schnieder, M., Nikoo, M., Vigo, D., Jang, K., Demlova, R., & Krausz, M. (2019). Mobile Apps for Medication Management: Review and Analysis. JMIR mHealth and uHealth, 7(9), e13608. https://doi.org/10.2196/13608
9. Brickwood, K.-J., Watson, G., O’Brien, J., & Williams, A. D. (2019). Consumer-Based Wearable Activity Trackers Increase Physical Activity Participation: Systematic Review and Meta-Analysis. JMIR mHealth and uHealth, 7(4), e11819. https://doi.org/10.2196/11819
10. Faiola, A., Papautsky, E. L., & Isola, M. (2019). Empowering the Aging with Mobile Health: A mHealth Framework for Supporting Sustainable Healthy Lifestyle Behavior. Current Problems in Cardiology, 44(8), 232–266. https://doi.org/10.1016/j.cpcardiol.2018.06.003
11. Hamine, S., Gerth-Guyette, E., Faulx, D., Green, B. B., & Ginsburg, A. S. (2015). Impact of mHealth Chronic Disease Management on Treatment Adherence and Patient Outcomes: A Systematic Review. Journal of Medical Internet Research, 17(2), e52. https://doi.org/10.2196/jmir.3951
12. Stevenson, J. K., Campbell, Z. C., Webster, A. C., Chow, C. K., Tong, A., Craig, J. C., Campbell, K. L., & Lee, V. W. (2019). E-Health interventions for people with chronic kidney disease. Cochrane Database of Systematic Reviews, 2019(8). https://doi.org/10.1002/14651858.CD012379.pub2
13. Pouls, B. P. H., Vriezekolk, J. E., Bekker, C. L., Linn, A. J., van Onzenoort, H. A. W., Vervloet, M., van Dulmen, S., & van den Bemt, B. J. F. (2021). Effect of Interactive e-Health Interventions on Improving Medication Adherence in Adults With Long-Term Medication: Systematic Review. Journal of Medical Internet Research, 23(1), e18901. https://doi.org/10.2196/18901
14. Wong, Z. S., Siy, B., Da Silva Lopes, K., & Georgiou, A. (2020). Improving Patients’ Medication Adherence and Outcomes in Nonhospital Settings Through e-Health: Systematic Review of Randomized Controlled Trials. Journal of Medical Internet Research, 22(8), e17015. https://doi.org/10.2196/17015
15. Latulippe, K., Hamel, C., & Giroux, D. (2017). Social Health Inequalities and e-Health: A Literature Review With Qualitative Synthesis of Theoretical and Empirical Studies. Journal of Medical Internet Research, 19(4), e136. https://doi.org/10.2196/jmir.6731
16. Hansen, A. H., Bradway, M., Broz, J., Claudi, T., Henriksen, Ø., Wangberg, S. C., & Årsand, E. (2019). Inequalities in the Use of e-Health Between Socioeconomic Groups Among Patients With Type 1 and Type 2 Diabetes: Cross-Sectional Study. Journal of Medical Internet Research, 21(5), e13615. https://doi.org/10.2196/13615
17. Herndon, J. B., Chaney, M., & Carden, D. (2011). Health Literacy and Emergency Department Outcomes: A Systematic Review. Annals of Emergency Medicine, 57(4), 334–345. https://doi.org/10.1016/j.annemergmed.2010.08.035
18. Bommakanti, K. K., Smith, L. L., Liu, L., Do, D., Cuevas-Mota, J., Collins, K., Munoz, F., Rodwell, T. C., & Garfein, R. S. (2020). Requiring smartphone ownership for mHealth interventions: Who could be left out? BMC Public Health, 20(1), 81. https://doi.org/10.1186/s12889-019-7892-9
19. van Boven, J. F., Tsiligianni, I., Potočnjak, I., Mihajlović, J., Dima, A. L., Nabergoj Makovec, U., Ágh, T., Kardas, P., Ghiciuc, C. M., Petrova, G., Bitterman, N., Kamberi, F., Culig, J., & Wettermark, B. (2021). European Network to Advance Best Practices and Technology on Medication Adherence: Mission Statement. Frontiers in Pharmacology, 12. https://doi.org/10.3389/fphar.2021.748702
20. Côté, J., Fortin, M.-C., Auger, P., Rouleau, G., Dubois, S., Boudreau, N., Vaillant, I., & Gélinas-Lemay, É. (2018). Web-Based Tailored Intervention to Support Optimal Medication Adherence Among Kidney Transplant Recipients: Pilot Parallel-Group Randomized Controlled Trial. JMIR Formative Research, 2(2), e14. https://doi.org/10.2196/formative.9707
21. DeVito Dabbs, A., Song, M. K., Myers, B. A., Li, R., Hawkins, R. P., Pilewski, J. M., Bermudez, C. A., Aubrecht, J., Begey, A., Connolly, M., Alrawashdeh, M., & Dew, M. A. (2016). A Randomized Controlled Trial of a Mobile Health Intervention to Promote Self-Management After Lung Transplantation. American Journal of Transplantation, 16(7), 2172–2180. https://doi.org/10.1111/ajt.13701
22. Hosseininasab, M., Jahangard-Rafsanjani, Z., Mohagheghi, A., Sarayani, A., Rashidian, A., Javadi, M., Ahmadvand, A., Hadjibabaie, M., & Gholami, K. (2014). Self-Monitoring of Blood Pressure for Improving Adherence to Antihypertensive Medicines and Blood Pressure Control: A Randomized Controlled Trial. American Journal of Hypertension, 27(11), 1339–1345. https://doi.org/10.1093/ajh/hpu062
23. Hashimoto, S., Brinke, A. T., Roldaan, A. C., van Veen, I. H., Moller, G. M., Sont, J. K., Weersink, E. J. M., van der Zee, J. S., Braunstahl, G.-J., Zwinderman, A. H., Sterk, P. J., & Bel, E. H. (2011). Internet-based tapering of oral corticosteroids in severe asthma: A pragmatic randomised controlled trial. Thorax, 66(6), 514–520. https://doi.org/10.1136/thx.2010.153411
24. Düsing, R., Handrock, R., Klebs, S., Tousset, E., & Vrijens, B. (2009). Impact of supportive measures on drug adherence in patients with essential hypertension treated with valsartan: The randomized, open-label, parallel group study VALIDATE. Journal of Hypertension, 27(4), 894–901. https://doi.org/10.1097/HJH.0b013e328323f9be
25. Henriksson, J., Tydén, G., Höijer, J., & Wadström, J. (2016). A Prospective Randomized Trial on the Effect of Using an Electronic Monitoring Drug Dispensing Device to Improve Adherence and Compliance. Transplantation, 100(1), 203–209. https://doi.org/10.1097/TP.0000000000000971
26. Jeong, J. Y., Jeon, J.-H., Bae, K.-H., Choi, Y.-K., Park, K.-G., Kim, J.-G., Won, K. C., Cha, B. S., Ahn, C. W., Kim, D. W., Lee, C. H., & Lee, I.-K. (2018). Smart Care Based on Telemonitoring and Telemedicine for Type 2 Diabetes Care: Multi-Center Randomized Controlled Trial. Telemedicine and e-Health, 24(8), 604–613. https://doi.org/10.1089/tmj.2017.0203
27. Bobrow, K., Farmer, A. J., Springer, D., Shanyinde, M., Yu, L.-M., Brennan, T., Rayner, B., Namane, M., Steyn, K., Tarassenko, L., & Levitt, N. (2016). Mobile Phone Text Messages to Support Treatment Adherence in Adults With High Blood Pressure (SMS-Text Adherence Support [StAR]). Circulation, 133(6), 592–600. https://doi.org/10.1161/CIRCULATIONAHA.115.017530
28. Abughosh, S., Wang, X., Serna, O., Esse, T., Mann, A., Masilamani, S., Holstad, M. M., Essien, E. J., & Fleming, M. (2017). A Motivational Interviewing Intervention by Pharmacy Students to Improve Medication Adherence. Journal of Managed Care & Specialty Pharmacy, 23(5), 549–560. https://doi.org/10.18553/jmcp.2017.23.5.549
29. Boyne, J. J., Vrijhoef, H. J. M., Spreeuwenberg, M., De Weerd, G., Kragten, J., & Gorgels, A. P. M. (2014). Effects of tailored telemonitoring on heart failure patients’ knowledge, self-care, self-efficacy and adherence: A randomized controlled trial. European Journal of Cardiovascular Nursing, 13(3), 243–252. https://doi.org/10.1177/1474515113487464
30. Choudhry, N. K., Isaac, T., Lauffenburger, J. C., Gopalakrishnan, C., Lee, M., Vachon, A., Iliadis, T. L., Hollands, W., Elman, S., Kraft, J. M., Naseem, S., Doheny, S., Lee, J., Barberio, J., Patel, L., Khan, N. F., Gagne, J. J., Jackevicius, C. A., Fischer, M. A., … Sequist, T. D. (2018). Effect of a Remotely Delivered Tailored Multicomponent Approach to Enhance Medication Taking for Patients With Hyperlipidemia, Hypertension, and Diabetes. JAMA Internal Medicine, 178(9), 1182. https://doi.org/10.1001/jamainternmed.2018.3189
31. Marek, K. D., Stetzer, F., Ryan, P. A., Bub, L. D., Adams, S. J., Schlidt, A., Lancaster, R., & O’Brien, A.-M. (2013). Nurse Care Coordination and Technology Effects on Health Status of Frail Older Adults via Enhanced Self-Management of Medication. Nursing Research, 62(4), 269–278. https://doi.org/10.1097/NNR.0b013e318298aa55
32. Claborn, K. R., Leffingwell, T. R., Miller, M. B., Meier, E., & Stephens, J. R. (2014). Pilot study examining the efficacy of an electronic intervention to promote HIV medication adherence. AIDS Care, 26(3), 404–409. https://doi.org/10.1080/09540121.2013.824534
33. Cizmic, A. D., Heilmann, R. M. F., Milchak, J. L., Riggs, C. S., & Billups, S. J. (2015). Impact of interactive voice response technology on primary adherence to bisphosphonate therapy: A randomized controlled trial. Osteoporosis International, 26(8), 2131–2136. https://doi.org/10.1007/s00198-015-3116-z
34. Márquez Contreras, E., Márquez Rivero, S., Rodríguez García, E., López-García-Ramos, L., Carlos Pastoriza Vilas, J., Baldonedo Suárez, A., Gracia Diez, C., Gil Guillén, V., & Martell Claros, N. (2019). Specific hypertension smartphone application to improve medication adherence in hypertension: A cluster-randomized trial. Current Medical Research and Opinion, 35(1), 167–173. https://doi.org/10.1080/03007995.2018.1549026
35. Mira, J. J., Navarro, I., Botella, F., Borrás, F., Nuño-Solinís, R., Orozco, D., Iglesias-Alonso, F., Pérez-Pérez, P., Lorenzo, S., & Toro, N. (2014). A Spanish Pillbox App for Elderly Patients Taking Multiple Medications: Randomized Controlled Trial. Journal of Medical Internet Research, 16(4), e99. https://doi.org/10.2196/jmir.3269
36. Montalescot, G., Brotons, C., Cosyns, B., Crijns, H. J., D’Angelo, A., Drouet, L., Eberli, F., Lane, D. A., Besse, B., Chan, A., Vicaut, E., & Darius, H. (2020). Educational Impact on Apixaban Adherence in Atrial Fibrillation (the AEGEAN STUDY): A Randomized Clinical Trial. American Journal of Cardiovascular Drugs, 20(1), 61–71. https://doi.org/10.1007/s40256-019-00356-2
37. Morawski, K., Ghazinouri, R., Krumme, A., Lauffenburger, J. C., Lu, Z., Durfee, E., Oley, L., Lee, J., Mohta, N., Haff, N., Juusola, J. L., & Choudhry, N. K. (2018). Association of a Smartphone Application With Medication Adherence and Blood Pressure Control. JAMA Internal Medicine, 178(6), 802. https://doi.org/10.1001/jamainternmed.2018.0447
38. O’Connor, P. J., Schmittdiel, J. A., Pathak, R. D., Harris, R. I., Newton, K. M., Ohnsorg, K. A., Heisler, M., Sterrett, A. T., Xu, S., Dyer, W. T., Raebel, M. A., Thomas, A., Schroeder, E. B., Desai, J. R., & Steiner, J. F. (2014). Randomized Trial of Telephone Outreach to Improve Medication Adherence and Metabolic Control in Adults With Diabetes. Diabetes Care, 37(12), 3317–3324. https://doi.org/10.2337/dc14-0596
39. Reese, P. P., Bloom, R. D., Trofe-Clark, J., Mussell, A., Leidy, D., Levsky, S., Zhu, J., Yang, L., Wang, W., Troxel, A., Feldman, H. I., & Volpp, K. (2017). Automated Reminders and Physician Notification to Promote Immunosuppression Adherence Among Kidney Transplant Recipients: A Randomized Trial. American Journal of Kidney Diseases, 69(3), 400–409. https://doi.org/10.1053/j.ajkd.2016.10.017
40. Rinfret, S., Lussier, M.-T., Peirce, A., Duhamel, F., Cossette, S., Lalonde, L., Tremblay, C., Guertin, M.-C., LeLorier, J., Turgeon, J., & Hamet, P. (2009). The Impact of a Multidisciplinary Information Technology–Supported Program on Blood Pressure Control in Primary Care. Circulation: Cardiovascular Quality and Outcomes, 2(3), 170–177. https://doi.org/10.1161/CIRCOUTCOMES.108.823765
41. Kamal, A. K., Khalid, W., Muqeet, A., Jamil, A., Farhat, K., Gillani, S. R. A., Zulfiqar, M., Saif, M., Muhammad, A. A., Zaidi, F., Mustafa, M., Gowani, A., Sharif, S., Bokhari, S. S., Tai, J., Rahman, N., Sultan, F. A. T., Sayani, S., & Virani, S. S. (2018). Making prescriptions “talk” to stroke and heart attack survivors to improve adherence: Results of a randomized clinical trial (The Talking Rx Study). PLOS ONE, 13(12), e0197671. https://doi.org/10.1371/journal.pone.0197671
42. Kessler, J. B., Troxel, A. B., Asch, D. A., Mehta, S. J., Marcus, N., Lim, R., Zhu, J., Shrank, W., Brennan, T., & Volpp, K. G. (2018). Partners and Alerts in Medication Adherence: A Randomized Clinical Trial. Journal of General Internal Medicine, 33(9), 1536–1542. https://doi.org/10.1007/s11606-018-4389-7
43. Kooij, M. J., Heerdink, E. R., van Dijk, L., van Geffen, E. C. G., Belitser, S. V., & Bouvy, M. L. (2016). Effects of Telephone Counseling Intervention by Pharmacists (TelCIP) on Medication Adherence; Results of a Cluster Randomized Trial. Frontiers in Pharmacology, 7(AUG), 1–11. https://doi.org/10.3389/fphar.2016.00269
44. Kooy, M. J., van Wijk, B. L. G., Heerdink, E. R., de Boer, A., & Bouvy, M. L. (2013). Does the use of an electronic reminder device with or without counseling improve adherence to lipid-lowering treatment? The results of a randomized controlled trial. Frontiers in Pharmacology, 4(May), 1–11. https://doi.org/10.3389/fphar.2013.00069
45. Levine, D., Torabi, J., Choinski, K., Rocca, J. P., & Graham, J. A. (2019). Transplant surgery enters a new era: Increasing immunosuppressive medication adherence through mobile apps and smart watches. The American Journal of Surgery, 218(1), 18–20. https://doi.org/10.1016/j.amjsurg.2019.02.018
46. Liu, W.-T., Huang, C.-D., Wang, C.-H., Lee, K.-Y., Lin, S.-M., & Kuo, H.-P. (2011). A mobile telephone-based interactive self-care system improves asthma control. European Respiratory Journal, 37(2), 310–317. https://doi.org/10.1183/09031936.00000810
47. Willems, D. C. M., Joore, M. A., Hendriks, J. J. E., Nieman, F. H. M., Severens, J. L., & Wouters, E. F. M. (2008). The effectiveness of nurse-led telemonitoring of asthma: Results of a randomized controlled trial. Journal of Evaluation in Clinical Practice, 14(4), 600–609. https://doi.org/10.1111/j.1365-2753.2007.00936.x
48. Young, L., Hertzog, M., & Barnason, S. (2016). Effects of a home-based activation intervention on self-management adherence and readmission in rural heart failure patients: The PATCH randomized controlled trial. BMC Cardiovascular Disorders, 16(1), 176. https://doi.org/10.1186/s12872-016-0339-7
49. Márquez Contreras, E., Márquez Rivero, S., Rodríguez García, E., López-García-Ramos, L., Carlos Pastoriza Vilas, J., Baldonedo Suárez, A., Gracia Diez, C., Gil Guillén, V., & Martell Claros, N. (2019). Specific hypertension smartphone application to improve medication adherence in hypertension: A cluster-randomized trial. Current Medical Research and Opinion, 35(1), 167–173. https://doi.org/10.1080/03007995.2018.1549026
50. Santschi, V., Rodondi, N., Bugnon, O., & Burnier, M. (2008). Impact of electronic monitoring of drug adherence on blood pressure control in primary care: A cluster 12-month randomised controlled study. Em European Journal of Internal Medicine(Vol. 19, Número 6, pp. 427–434). https://doi.org/10.1016/j.ejim.2007.12.007
51. Svendsen, M. T., Andersen, F., Andersen, K. H., Pottegård, A., Johannessen, H., Möller, S., August, B., Feldman, S. R., & Andersen, K. E. (2018). A smartphone application supporting patients with psoriasis improves adherence to topical treatment: A randomized controlled trial. British Journal of Dermatology, 179(5), 1062–1071. https://doi.org/10.1111/bjd.16667
52. Reese, P. P., Kessler, J. B., Doshi, J. A., Friedman, J., Mussell, A. S., Carney, C., Zhu, J., Wang, W., Troxel, A., Young, P., Lawnicki, V., Rajpathak, S., & Volpp, K. (2016). Two Randomized Controlled Pilot Trials of Social Forces to Improve Statin Adherence among Patients with Diabetes. Journal of General Internal Medicine, 31(4), 402–410. https://doi.org/10.1007/s11606-015-3540-y
53. Wakefield, B. J., Holman, J. E., Ray, A., Scherubel, M., Adams, M. R., Hills, S. L., & Rosenthal, G. E. (2012). Outcomes of a home tele-Health intervention for patients with diabetes and hypertension. Telemedicine and e-Health, 18(8), 575–579. https://doi.org/10.1089/tmj.2011.0237
54. Wald, D. S., Bestwick, J. P., Raiman, L., Brendell, R., & Wald, N. J. (2014). Randomised trial of text messaging on adherence to cardiovascular preventive treatment (INTERACT Trial). PLoS ONE, 9(12), 1–9. https://doi.org/10.1371/journal.pone.0114268
55. Abughosh, S. M., Wang, X., Serna, O., Henges, C., Masilamani, S., James Essien, E., Chung, N., & Fleming, M. (2016). A Pharmacist Telephone Intervention to Identify Adherence Barriers and Improve Adherence Among Nonadherent Patients with Comorbid Hypertension and Diabetes in a Medicare Advantage Plan. Journal of Managed Care & Specialty Pharmacy, 22(1), 63–73. https://doi.org/10.18553/jmcp.2016.22.1.63
56. Vollmer, W. M., Owen-Smith, A. A., Tom, J. O., Laws, R., Ditmer, D. G., Smith, D. H., Waterbury, A. C., Schneider, J. L., Yonehara, C. H., Williams, A., Vupputuri, S., & Rand, C. S. (2014). Improving adherence to cardiovascular disease medications with information technology. The American journal of managed care, 20(11 Spec No. 17), SP502-10.
57. Sherrard, H., Struthers, C., Kearns, S. A., Wells, G., Chen, L., & Mesana, T. (2009). Using technology to create a medication safety net for cardiac surgery patients: A nurse-led randomized control trial. Canadian journal of cardiovascular nursing = Journal canadien en soins infirmiers cardio-vasculaires, 19(3), 9–15.
58. Stacy, J. N., Schwartz, S. M., Ershoff, D., & Shreve, M. S. (2009). Incorporating Tailored Interactive Patient Solutions Using Interactive Voice Response Technology to Improve Statin Adherence: Results of a Randomized Clinical Trial in a Managed Care Setting. Population Health Management, 12(5), 241–254. https://doi.org/10.1089/pop.2008.0046
59. Jerant, A. F., Azari, R., Martinez, C., & Nesbitt, T. S. (2003). A Randomized Trial of Telenursing to Reduce Hospitalization for Heart Failure: Patient-Centered Outcomes and Nursing Indicators. Home Health Care Services Quarterly, 22(1), 1–20. https://doi.org/10.1300/J027v22n01_01
60. Kamal, A. K., Shaikh, Q., Pasha, O., Azam, I., Islam, M., Memon, A. A., Rehman, H., Akram, M. A., Affan, M., Nazir, S., Aziz, S., Jan, M., Andani, A., Muqeet, A., Ahmed, B., & Khoja, S. (2015). A randomized controlled behavioral intervention trial to improve medication adherence in adult stroke patients with prescription tailored Short Messaging Service (SMS)-SMS4Stroke study. BMC Neurology, 15(1), 212. https://doi.org/10.1186/s12883-015-0471-5
61. Ducoulombier, V., Luraschi, H., Forzy, G., Vandecandelaere, M., & Houvenagel, E. (2015). Contribution of phone follow-up to improved adherence to oral osteoporosis treatment. Am J Pharm Benefits, 7(3), e81–e89.
62. Bosworth, H. B., Powers, B. J., Olsen, M. K., McCant, F., Grubber, J., Smith, , Gentry, P. W., Rose, C., Houtven, C. Van, Wang, V., Goldstein, M. K., & Oddone, E. Z. (2011). Home Blood Pressure Management and Improved Blood Pressure Control. Archives of Internal Medicine, 171(13), 1173–1180. https://doi.org/10.1001/archinternmed.2011.276
63. Kim, Y., Wineinger, Nathan. E., & Steinhubl, Steven. R. (2016). The Influence of Wireless Self-Monitoring Program on the Relationship Between Patient Activation and Health Behaviors, Medication Adherence, and Blood Pressure Levels in Hypertensive Patients: A Substudy of a Randomized Controlled Trial. Journal of Medical Internet Research, 18(6), e116. https://doi.org/10.2196/jmir.5429
64. Vicente, R., & Madden, G. (2017). Assessing e-Health skills across Europeans. Health Policy and Technology, 6(2), 161–168. https://doi. org/10.1016/j.hlpt.2017.04.001
65. Norman, C. D., & Skinner, H. A. (2006). EHEALS: The e-Health Literacy Scale. Journal of Medical Internet Research, 8(4), e27. https://doi. org/10.2196/jmir.8.4.e27
66. Butler, , & Savalli, S. J. E. (2021). The Rising Implementation of Digital Health Platforms and Its Associated Inequities Among Patients with Mental Disorders: A Literature Review. Undergraduate Research in Natural and Clinical Science and Technology (URNCST) Journal, 5(1), 1–10. https://doi.org/10.26685/urncst.212
67. Mangin, D., Parascandalo, J., Khudoyarova, O., Agarwal, G., Bismah, V., & Orr, S. (2019). Multimorbidity, e-Health and implications for equity: A cross-sectional survey of patient perspectives on e-Health. BMJ Open, 9(2), e023731. https://doi.org/10.1136/bmjopen-2018-023731
68. Maffoni, M., Traversoni, S., Costa, E., Midão, L., Kardas, P., Kurczewska-Michalak, M., & Giardini, A. (2020). Medication adherence in the older adults with chronic multimorbidity: A systematic review of qualitative studies on patient’s European Geriatric Medicine, 11(3), 369–381. https://doi.org/10.1007/s41999-020-00313-2
69. Wilson, J., Heinsch, M., Betts, D., Booth, D., & Kay-Lambkin, F. (2021). Barriers and facilitators to the use of e-Health by older adults: A scoping BMC Public Health, 21(1), 1556. https://doi.org/10.1186/s12889-021-11623-w
70. Gregório, J., Reis, L., Peyroteo, M., Maia, M., Mira da Silva, M., & Lapão, L. (2021). The role of Design Science Research Methodology in developing pharmacy e-Health services. Research in Social and Administrative Pharmacy, 17(12), 2089–2096. https://doi.org/10.1016/j. sapharm.2021.05.016
71. Scandurra, I., & Sjölinder, M. (2013). Participatory Design With Seniors: Design of Future Services and Iterative Refinements of Interactive e-Health Services for Old Citizens. Medicine 2.0, 2(2), e12. https://doi.org/10.2196/med20.2729
72. Neves, A. L., Jácome, C., Taveira-Gomes, T., Pereira, A. M., Almeida, R., Amaral, R., Alves-Correia, M., Mendes, S., Chaves-Loureiro, C., Valério, , Lopes, C., Carvalho, J., Mendes, A., Ribeiro, C., Prates, S., Ferreira, J. A., Teixeira, M. F., Branco, J., Santalha, M., … Almeida Fonseca, J. (2021). Determinants of the Use of Health and Fitness Mobile Apps by Patients With Asthma: Secondary Analysis of Observational Studies. Journal of Medical Internet Research, 23(9), e25472. https://doi.org/10.2196/25472
73. Azzopardi-Muscat, N., & Sørensen, K. (2019). Towards an equitable digital public health era: Promoting equity through a health literacy European Journal of Public Health, 29(Supplement_3), 13–17. https://doi.org/10.1093/eurpub/ckz166
74. Gordon, P., & Hornbrook, M. C. (2016). Differences in Access to and Preferences for Using Patient Portals and Other e-Health Technologies Based on Race, Ethnicity, and Age: A Database and Survey Study of Seniors in a Large Health Plan. Journal of Medical Internet Research, 18(3), e50. https://doi.org/10.2196/jmir.5105
75. Olu, O., Muneene, D., Bataringaya, J. E., Nahimana, M.-R., Ba, H., Turgeon, Y., Karamagi, H. C., & Dovlo, D. (2019). How Can Digital Health Technologies Contribute to Sustainable Attainment of Universal Health Coverage in Africa? A Perspective. Frontiers in Public Health, 7. https:// doi.org/10.3389/fpubh.2019.00341
76. Damschroder, L. J., Aron, D. C., Keith, R. E., Kirsh, S. R., Alexander, J. A., & Lowery, J. C. (2009). Fostering implementation of health services research findings into practice: A consolidated framework for advancing implementation science. Implementation Science, 4(1), 50. https:// org/10.1186/1748-5908-4-50
77. Luetsch, K., Maidment, I., Twigg, M., & Rowett, D. (2021). Realist research to inform pharmacy practice and policy. Research in Social and Administrative Pharmacy, 17(12), 2075–2081. https://doi.org/10.1016/j.sapharm.2021.06.026
