![]()
Biopharmaceutical Sciences, Biomed Biopharm Res., 2021; 18(2):274-290
doi: 10.19277/bbr.18.2.272; [+] PDF version here; [+] Portuguese html here
Cecropia pachystachya protection against preproIAPP cytotoxicity is independent of Ca2+ homeostasis: lessons learned using a novel yeast model of preproIAPP-induced Ca2+ intracellular dysregulation
Sofia Ferreira 1, Rejane G. Tavares 1,2, Ana F. Raimundo 3,4,5, Francieli M. Stefanello 6, Flavio H. Reginatto 7, Nuno Saraiva 1, Regina Menezes 1,3,5*
1 CBIOS - Center for Biosciences & Health Technologies, Universidade Lusófona de Humanidades e Tecnologias, Campo Grande 376, 1749-024 Lisboa, Portugal;
2 Programa de Pós-Graduação em Nutrição e Alimentos, Faculdade de Nutrição, Universidade Federal de Pelotas, Pelotas, RS, Brasil;
3 iBET - Instituto de Biologia Experimental e Tecnológica, Apartado 12, 2781-901 Oeiras, Portugal;
4 ITQB-NOVA, Instituto de Tecnologia Química e Biológica António Xavier, Universidade Nova de Lisboa, Av. da República, 2781-157 Oeiras, Portugal;
5 CEDOC, NOVA Medical School|Faculdade de Ciências Médicas, Universidade NOVA de Lisboa, Campo dos Mártires da Pátria, 130, 1169-056 Lisboa, Portugal;
6 Programa de Pós-Graduação em Bioquímica e Bioprospecção, Centro de Ciências Químicas, Farmacêuticas e de Alimentos, Universidade Federal de Pelotas, Pelotas, RS, Brasil;
7 Programa de Pós-Graduação em Farmácia, Centro de Ciências da Saúde, Universidade Federal de Santa Catarina, Florianópolis, SC, Brasil
*corresponding author:
Abstract
Despite the progresses in therapeutics, type 2 diabetes is still an epidemic with alarming numbers worldwide. New strategies for prevention and treatments are imperative. A venue of research with potential is the aggregation of Islet Amyloid PolyPeptide (IAPP), a contributor to pancreatic β-cell dysfunction. To address the impact of IAPP on calcium (Ca2+) signalling, we developed a dual reporter yeast model independently expressing preproIAPP-GFP (ppIAPP) and encoding the lacZ gene under the control of Crz1-recognition elements. In this reporter system, ppIAPP induced Crz1 hyperactivation, a yeast transcription factor activated by the Ca2+/calmodulin/calcineurin pathway, which was followed by the increase in β-galactosidase activity. Encouraged by the reported healthy effects of Urticaceae plants against metabolic disturbances, we tested the protective potential of Cecropia pachystachya against IAPP-induced cytotoxicity using the newly designed yeast model. Although C. pachystachya extract exerted no beneficial effects towards the prevention of ppIAPP cytotoxicity, treatment with C. pachystachya enriched C-glycosyl flavonoid fraction (EFF-Cp) significantly improved viability of ppIAPP-expressing cells. Potential bioactivities of C. pachystachya extract and EFF-Cp towards the restoration of Ca2+ homeostasis disrupted by ppIAPP expression were also assessed. Neither prevented Crz1 hyperactivation, suggesting that the EFF-Cp-induced protection against ppIAPP toxicity was mediated by Ca2+-independent mechanisms.
Keywords: Amylin, Calcium signaling; Cecropia pachystachya; Diabetes; Islet Amyloid Polypeptide (IAPP)
Received: 02/12/2021; Accepted: 31/12/2021
Introduction
Diabetes is the most prevalent metabolic disorder representing a major social and economic burden worldwide. Multiple factors contribute to the onset and progression of diabetes. The main pathological features of the disease include high blood glucose levels due to defective insulin sensitivity or insufficient insulin production/secretion by dysfunctional pancreatic β-cells (1). One undeniable contributor to β-cell damage in diabetes is the aggregation of Islet Amyloid Polypeptide (IAPP), also called amylin, a hormone co-produced with insulin by β-cells in response to blood glucose levels (2). During its synthesis, immature IAPP forms are sequentially processed by hormone convertases to yield the mature and functional 37-amino acid IAPP molecule which acts synergistically with insulin to stabilize glucose homeostasis. In pre-diabetes/diabetes conditions, the boost in insulin production induced by hyperglycaemia is accompanied by an exacerbated production of IAPP. This leads to the overload of β-cell processing machinery and the intracellular accumulation of highly amyloidogenic immature IAPP species (3). Intracellular toxic oligomers of IAPP have been described to impair virtually all β-cell functions. They affect cell proliferation, increase Endoplasmic Reticulum (ER) stress, impair autophagy and mitochondrial function, dysregulate the Unfolded Protein Response (UPR), exacerbate local inflammation, among other effects (4–7). Together, these events culminate in β-cell injury and ultimately contribute to the disease development.
Amyloid peptides have been reported to induce β-cell death through apoptosis, which may be a consequence of disruption of calcium (Ca2+) homeostasis (8-9). IAPP oligomers interfere with cellular membrane structure and dynamics, serving as the origin of ionic pores that allow the uncontrolled entry of Ca2+ ions into the intracellular environment, thus leading to the activation of an apoptosis cascade (10-11). Importantly, dysregulation of β-cell Ca2+ levels is associated with insulin resistance, a key feature of diabetes (12). Alterations in cytosolic free Ca2+ levels play a crucial role in the control of physiological insulin release from pancreatic β-cells. Upon glucose metabolism, ATP levels increase, inducing the inhibition of ATP-regulated potassium channels. Consequently, the plasma membrane depolarizes, producing an electrical change that opens voltage-gated Ca2+ channels. The influx of Ca2+ ions triggers the release of secretory granules containing insulin which are externalized by exocytosis. Altered steady-state Ca2+ levels have been observed in a wide range of cell types and tissues (e.g., platelets, cardiomyocytes, β-cells, muscle, kidney, liver, adipocytes, osteoblasts, among others) from Type 2 Diabetes Mellitus (T2DM) patients and animal models, indicating that this serves as a fundamental factor in diabetic state (12-13).
In the last decades, several studies have reported the potential of phytochemicals from natural sources to prevent or control metabolic alterations associated with chronic diseases. Some plants of the genus Cecropia (Urticaceae), native to Central and South America and known as “embaúba” in popular culture (14), are an example of this case. In particular, Cecropia pachystachya leaves have been widely used as hypoglycaemic, diuretic, anti-inflammatory, and antioxidant (14–19). In addition to their properties against metabolic dysfunctions, in vivo studies using enriched C-glycosyl flavonoid fractions and/or aqueous extracts from C. pachystachya leaves have shown effects within the central nervous system (20-22), whereas in vitro, C. pachystachya extracts were shown to be cytotoxic for several types of cancer cells (15-19). The pharmacological properties of C. pachystachya have been correlated to their content of phenolic compounds, majorly chlorogenic acid, isoorientin, orientin, isovitexin, and isoquercitrin (21-22).
In this study, we further addressed the protective action of C. pachystachya extracts and fractions using Saccharomyces cerevisiae models of IAPP-induced toxicity (23) as a potential approach to target pathological processes of diabetes.
Materials and Methods
Plant material
Aerial parts of C. pachystachya Trécul leaves were collected in Torres, RS, Brazil. A voucher specimen (ICN 150025) was deposited in the Herbarium of the Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre, Brazil. The access to the Brazilian biodiversity was registered in the National System for Genetic Heritage Management and Associated Traditional Knowledge (SISGEN) under protocol AC5A37.
Aqueous extract preparation
The leaves of C. pachystachya were air-dried (35–40◦C) for three days and then extracted by infusion (1:10, plant/distilled water) for 30 min, filtered, freeze-dried, and stored at −20◦C until use (22).
Enriched fractions preparation
The enriched C-glycosyl flavonoid fraction (EFF-Cp) was obtained as previously described by Ortmann et al. (2016) (21). The dried plant material was submitted to extraction with ethanol 20% (5% plant:solvent ratio, w/v; 9500 rpm; 5 min) and the extract was partitioned with n-butanol, yielding the n-butanolic fraction. The butanolic fraction was dried under reduced pressure and stirred with Amberlite® XAD-16 resin (Sigma-Aldrich; St Louis, MO, USA). The resin was then stirred again in the presence of methanol for 30 min, yielding the EFF-Cp.
Chemical characterization of extracts and fractions
The extract was chemically analyzed by HPLC/DAD as previously described by Costa et al. (2011) (24) and Ortmann et al (2016) (21), using a Perkin Elmer Series 200 HPLC with a Photo Diode Array Detector (PDA) (PerkinElmer, Shelton, CT, USA).
Yeast strains and plasmids
S. cerevisiae strains used in this study are listed in Table 1. The strain YAA5, which encodes the CDRE-lacZ reporter gene, was used for cell viability and β-galactosidase activity assays. The parental strain BY4742 and the strains YAA6 and YAA7 were used as negative controls for the β-galactosidase activity assays. The plasmids used are listed in Table 2. Yeast transformation procedures were carried out as indicated using the lithium acetate standard method (25).


Growth conditions
Synthetic dropout (SD)-2% glucose medium was used for the growth of cells transformed with the plasmids of interest. A pre-inoculum was prepared in SD-raffinose medium, and cultures were incubated overnight at 30°C under orbital shaking. Cultures were diluted in fresh medium and incubated under the same conditions until the optical density at 600 nm (OD600) reached log growth phase. The following equation was used to synchronize the cultures: ODi × Vi = ODf / [2(t/gt)] × Vf, where ODi is the initial optical density of the culture, Vi is the initial volume of culture, ODf is the final optical density of the culture, t is the time, gt is the generation time of the strain, and Vf is the final volume of culture. Cell cultures were diluted as indicated for each assay. In all experiments, induction of IAPP expression was carried out in SD-galactose medium. Media composition is detailed in Raimundo et al. (2020) (23).
Flow cytometry
Cell cultures were diluted to OD600 0.1 ± 0.01 in SD-galactose and incubated with 0, 50 and 100 µg/mL of EFF-Cp or C. pachystachya aqueous extract, at 30°C for 20 h under orbital agitation. Cells were incubated with propidium iodide (PI) (Merck, Darmstadt, DE) at a final concentration of 2.5 μg/ml for 30 min at 30°C under orbital agitation and protected from light. Flow cytometry was performed using a BD FACS Calibur Flow Cytometer (BD Biosciences, San Jose, CA, USA). Data acquisition and analysis were performed using CellQuest® (BD Biosciences, San Jose, CA, USA) and FlowJo® software (Tree Star Inc., San Carlos, CA, USA), respectively. A minimum of 50000 events were collected for each experiment. Cell doublets exclusion was performed based on Forward-A and -W scatter parameters.
β-galactosidase activity
The β-galactosidase assays to infer Ca2+ homeostasis were carried out as described by Garcia et al. (2016)(26). Briefly, cultures at OD600=0.5±0.05 were diluted in SD-galactose medium to OD600=0.1±0.01, transferred to a 96-well microplate and incubated with 0 and 50 µg/mL of EFF-Cp or C. pachystachya aqueous extract for 20 h at 30°C under orbital agitation. After this period, OD600 of cultures were measured by the transfer of 10 μL of cell suspensions to a new 96-well plate followed by addition of 20 μL Y-PER cell lysis reagent (Thermo Fisher Scientific Inc.–Life Technologies, USA). The plate was incubated for 20 min at 37◦C without agitation. 240 μL of lacZ buffer [8.5 g/L Na2HPO4 (Carl Roth GmbH & Co. KG, Karlsruhe, DE), 5.5 g/L NaH2PO4.H2O (Merck, Darmstadt, DE), 0.75 g/L KCl (Panreac, Bracelona, ES), 0.246 g/L MgSO4.7H2O (Merck, Darmstadt, DE) containing 4 mg/L o-nitrophenyl β-D-galactopyranoside (ONPG) (Sigma-Aldrich®, St Louis, MO, EUA)] was added to each well, and the plate was incubated at 30◦C for 2 h. The results were expressed as Miller units (26), applying the following equation, where V=volume of culture assayed in mL and t=reaction time in minutes:
Miller units = 1000×(OD420−1.75×OD550)/(t×V×OD600)
Statistical Analysis
Statistical analysis was expressed using Graphpad Prism 8 software. Data are mean ± SD of at least three independent biological replicates. One-way ANOVA with Dunnet’s multiple comparison test was performed to assess differences between the conditions. The t-test was used to access differences between the untreated p426 and ppIAPP-GFP conditions in the flow cytometry analysis. Two-way ANOVA with the Tukey’s or Sidak’s multiple comparison test was performed to access differences between the β-galactosidase assay conditions.
Results and Discussion
Development of a yeast model to address the effects of ppIAPP on Ca2+ signaling
Insulin secretion, β-cell survival, proliferation, and function are tightly controlled by Ca2+ dynamics. To determine the deleterious effect of IAPP on Ca2+ signaling, we took advantage of a previously described strategy we designed to recapitulate the molecular pathways of IAPP aggregation in the model organism Saccharomyces cerevisiae (23).
For this, we used the reporter strain YAA5 encoding the lacZ gene under the control of Crz1-recognition elements (Table 1), which is sensitive to Ca2+ intracellular levels, as the yeast transcription factor Crz1 is regulated by the calmodulin CaM/calcineurin (CaN) pathway (27). In a similar mechanism to the human orthologue NFAT, Crz1 activation is regulated by Ca2+ cytosolic concentrations. Slight oscillations in Ca2+ levels trigger CaM/CaN activation leading to Crz1 dephosphorylation, nuclear translocation, and stimulation of calcineurin-dependent response element (CDRE)-driven gene expression. Thus, the degree of Crz1 activation can be monitored through the measurement of β-galactosidase activity. The strains YAA6 and YAA7, devoid of CRZ1 and CNB1 (encoding the regulatory subunit of CaN complex), respectively, are used as control strains (Table 1).
The novel yeast models YAA5_ppIAPP-GFP, YAA6_ppIAPP-GFP and YAA7_ppIAPP-GFP consisted of YAA5, YAA6 and YAA7 strains, respectively, engineered to encode the most immature and toxic form of human IAPP (preproIAPP, ppIAPP) fused to the green fluorescent protein (GFP) and under the transcriptional control of a GAL1-inducible promoter (Figure 1A). As previously described by Raimundo et al. (2020) (23), the expression of such construct induces stronger toxic effects on yeast growth and cell viability than fusions of partially and fully processed forms of IAPP. For this reason, proIAPP-GFP and mature IAPP-GFP constructs were not included in the present study.
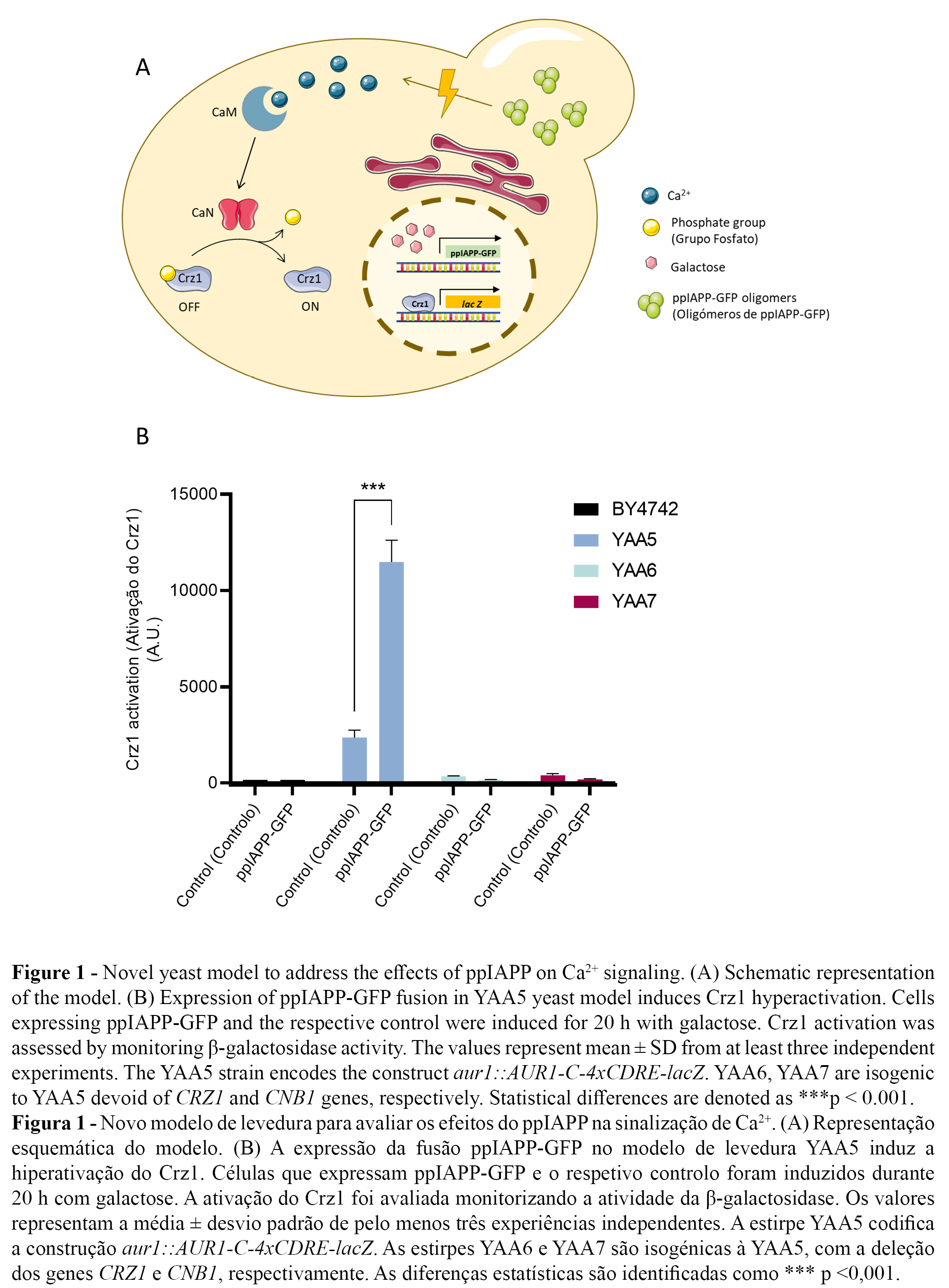
The expression of ppIAPP-GFP induced a strong activation of the CaM/CaN pathway in the YAA5 strain as indicated by the increase of Crz1-dependent β-galactosidase activity (Figure 1B). This is consistent with the reported effects of amyloid peptide on the disruption of Ca2+ homeostasis and dysregulated increase in cytosolic Ca2+ levels. By comparison, only a residual activity was observed in YAA6 and YAA7 cells. Likewise, in the parental strain BY4742 devoid of the CDRE-lacZ reporter construct, β-galactosidase activity was nearly undetectable (Figure 1B). These findings postulate our yeast model as a powerful platform for the bioprospection of compounds with potential to inhibit ppIAPP-induced Crz1 hyperactivation.
Chemical characterization of Cecropia pachystachya extract and fraction
The chromatographic profiles of C. pachystachya extract and fraction were similar to those described by Gazal et al. (2014) (22) as confirmed by quality control analysis indicating the presence of similar concentrations of major compounds present in the extract and fraction, namely chlorogenic acid, isoorientin, orientin, isovitexin and isoquercitrin (data not shown).
Cecropia pachystachya EFF-Cp fraction protects YAA5 yeast cells against ppIAPP-induced toxicity
The protective potential of phytochemicals from natural sources has been widely explored as alternative approaches against the metabolic alterations observed in many chronic diseases. More specifically, C. pachystachya bioactivity has been associated with the promotion of metabolic health by mechanisms including the regulation of glycaemic control, inflammation, and oxidative stress (14–17, 19).
S. cerevisiae is considered a robust primary drug-screening tool to filter for compounds with cytoprotective activity for further validation in more complex models (28–30). Among other features, this is possible because S. cerevisiae shares highly conserved molecular and cellular mechanisms with human counterparts.
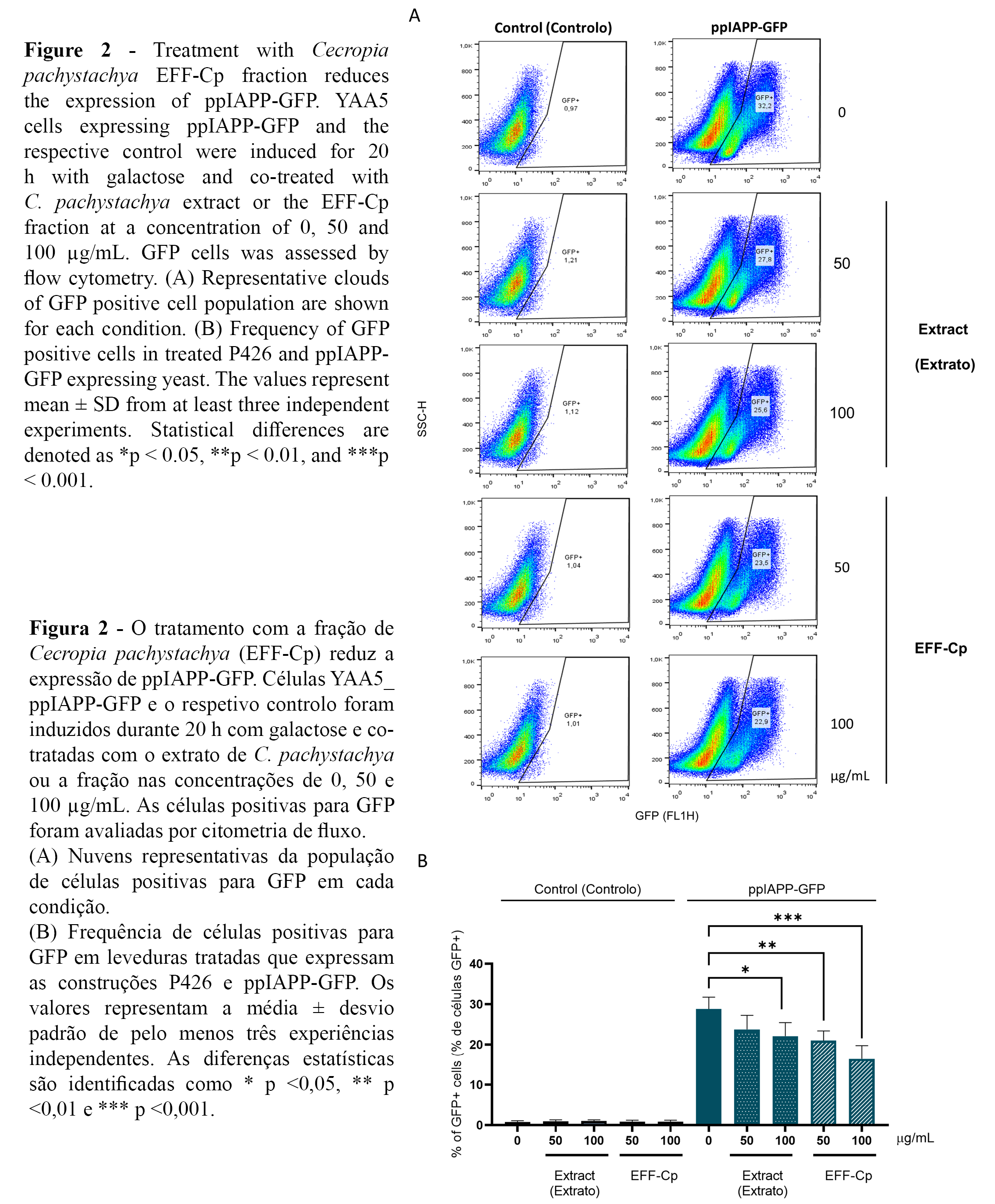
The reporter strain YAA5_ppIAPP-GFP was used to investigate further the potential protective action of C. pachystachya towards IAPP-induced cytotoxicity. ppIAPP-GFP expression was confirmed by flow cytometry, showing that nearly 30% of the total cell population displayed GFP signals in the control condition (Figure 2A, 2B). Interestingly, incubation with EFF-Cp induced a dose-dependent reduction in the percentage of GFP positive cell population in ppIAPP-GFP expressing yeast. However, further studies are needed to fully address the impact of EFF-Cp treatment on the expression ppIAPP-GFP fusion peptide.
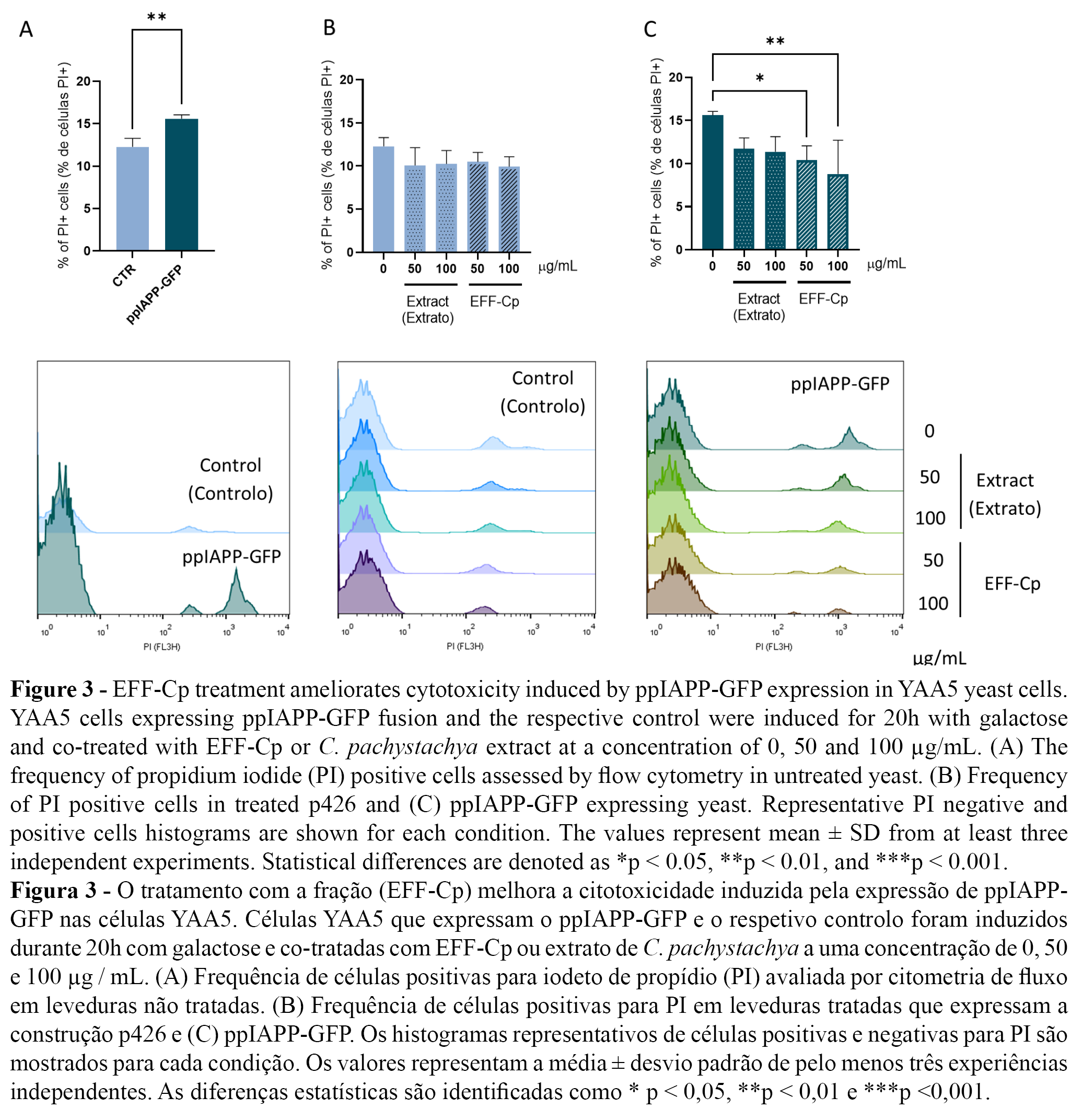
Next, membrane integrity was determined using PI staining as a measure of cell viability. The ppIAPP-GFP construct induced toxicity after 20 h incubation in the presence of galactose (Figure 3A). C. pachystachya extract and EFF-Cp did not affect cell viability in control cells (Figure 3B). Remarkably, EFF-Cp treatment significantly reduced the toxic effects mediated by ppIAPP-GFP with concentrations of 50 and 100 µg/mL, which can be deduced from the decrease of PI positive cells to values similar to the control condition (Figure 3C).
The protective action exerted by EFF-Cp may be attributed, at least in part, to chlorogenic acid and isoorientin, the major compounds identified in EFF-Cp. Both compounds have been previously associated with protective actions against diabetes. Concerning IAPP pathology, chlorogenic acid was shown to exert an inhibitory effect on human IAPP (hIAPP) oligomerization/fibril formation and to improve cell viability in pancreatic INS-1 cells exposed to hIAPP aggregates. By delaying the onset of secondary structure conversion of hIAPP from random coil to β-structure, it redirects the amyloidogenic molecules into off-pathway intermediates that are less toxic to the cells (31). Chlorogenic acid was also shown to decrease fasting blood glucose, lipid peroxidation (LPO) products, and glycosylated hemoglobin (HbA1C), and to increase plasma insulin, total hemoglobin, C-peptide, and glycogen in streptozotocin/nicotinamide (STZ/NA)-induced type 2 diabetic rats (32-33). It is currently argued that the hypoglycemic mechanisms of chlorogenic acid may be related to the inhibition of the glucose-6-phosphate-shifting enzyme, regulating the blood glucose balance in vivo. In addition, studies have shown that chlorogenic acid can activate AMP-activated protein kinase (AMPK), upregulate the gene expression of glucose transporter 4 (GLUT4) to stimulate the absorption of glucose in skeletal muscle, downregulate expression of glucose-6-phosphate-shifting enzyme, inhibit gluconeogenesis, and reduce fatty acids synthesis (34). In the liver, chlorogenic acid activity was also associated with reduced oxidative and ER stress, increased autophagy and improved mitochondrial activity (35). In a similar way, the major flavonoid compounds in EFF-Cp, orientin and its isomer isoorientin, have also been reported as potent regulators of oxidative stress. In addition, isoorientin may act to improve insulin sensitivity and limit lipid accumulation in diabetic animals and cultured adipocytes (36). Therefore, it is plausible to consider that protection conferred by EFF-Cp in our model system may be attributed to the concerted action of two main molecular events: (a) modulation of toxic IAPP aggregation and (b) mitigation of IAPP-induced redox imbalance.
Cecropia pachystachya bioactivity is unrelated to Ca2+ signalling
The cytosolic Ca2+ levels regulate a broad number of cellular processes, including metabolism, proliferation, transcription, and secretion. In β-cells, insulin granule exocytosis is tightly controlled by a finely tuned balance between Ca2+ influx and efflux pathways. Alterations in Ca2+ dynamics have been associated with impaired insulin secretion and β-cell dysfunction (37), thus representing an interesting target for a therapeutic approach.
Although there is no reported correlation between Cecropia pachystachya bioactivity and Ca2+ signalling, chlorogenic acid (one of the major components of EFF-Cp) was shown to suppress the dysregulated Ca2+ influx from endoplasmic reticulum and extracellular environment in endothelial cells exposed to a toxic phospholipid (38).
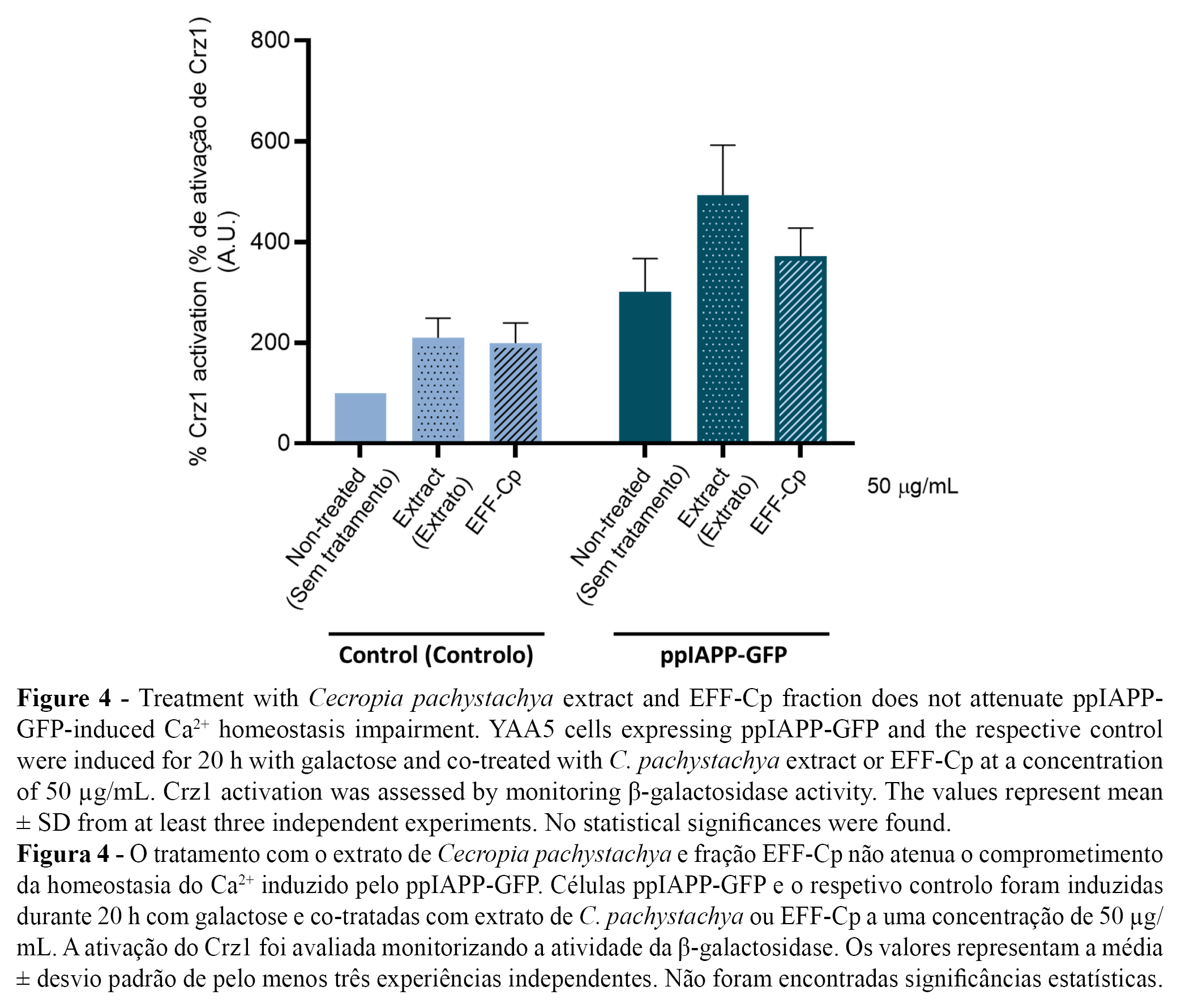
Taking advantage of the developed yeast-based reporter assay, we used YAA5 cells expressing control and ppIAPP-GFP constructs as “in vivo test-tubes” to screen the bioactivity of C. pachystachya extract and EFF-Cp towards the modulation of Ca2+ signalling and consequent Crz1 activation. Since treatment with the lower concentration (50 µg/mL) of EFF-Cp significantly protected cells against ppIAPP-GFP toxicity, we selected this concentration for further tests. The incubation of YAA5_ppIAPP-GFP cells with C. pachystachya extract did not affect β-galactosidase activity (Figure 4), which is in accordance with its ineffectiveness to protect cells against ppIAPP-GFP-induced toxicity in the viability assay. Similarly, in ppIAPP-GFP expressing cells, no alterations on Crz1 activation were observed after treatment with EFF-Cp. Since EFF-Cp is not able to inhibit Crz1 hyperactivation, we hypothesize that it might be conferring protection against deleterious effects of ppIAPP by modulating alternative molecular mechanisms that do not include CaM/CaN pathway. Additional studies are needed to fully comprehend the molecular and cellular mode of action underlying EFF-Cp protective effect.
Conclusions
With the development of a ppIAPP-expressing yeast model that acts as a sensor of intracellular Ca2+ levels, our study provided new insights about the interference of ppIAPP on Ca2+ signaling. Overexpression of ppIAPP induced the hyperactivation of Crz1 activity, which allowed us to identify Ca2+ signaling dysregulation as one of the pathological mechanisms underlying ppIAPP cytotoxicity. Importantly, such findings also offered a valuable high-throughput yeast-based reporter assay to screen for compounds preventing the deleterious effects of ppIAPP on Ca2+ dysregulation. Our study shows that the effectiveness of EFF-Cp to improve cell viability was not mediated by changes in Ca2+ signaling, suggesting that other mechanisms might be associated with this protective response. Noteworthy, EFF-Cp chemical composition is enriched in chlorogenic acid and isoorientin, two secondary metabolites exerting known protective properties towards diabetic complications and IAPP pathological effects. Based on the reported evidence, it seems plausible to hypothesize that cellular protection mediated by EFF-Cp may include the mitigation of oxidative stress, ER stress, mitochondrial damage, and autophagy. Further studies are needed to unravel the molecular mechanisms underlying EFF-Cp bioactivity.
Authors contributions statement
RM and RT conceived and designed the experiments. SF performed the experiments. SF, RT, AFR, FMS, FHR, NS and RM analyzed the data and wrote the paper. All authors contributed to the article and approved the submitted version.
Acknowledgements
This study was funded by Fundação para a Ciência e Tecnologia (FCT)/Ministério da Ciência e do Ensino Superior, grant numbers PTDC/BIA-MOL/31104/2017 (RM) and UIDB/04567/2020 and UIDP/ 04567/2020 (CBIOS). iNOVA4Health Research Unit (LISBOA—01–0145—FEDER—007344), which is cofunded by FCT/Ministério da Ciência e do Ensino Superior, through national funds, and by FEDER under the PT2020 Partnership Agreement, is also acknowledged. Authors would like to acknowledge FCT for the financial support of AFR (PD/BD/135504/2018); SF (UI/BD/151421/2021), and RM (CEEC/04567/CBIOS/2020).
Conflict of interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Kahn, S. E., Cooper, M. E., & Del Prato, S. (2014). Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet (London, England), 383(9922), 1068–1083. doi: 10.1016/S0140-6736(13)62154-6
2. Westermark, P., Andersson, A., & Westermark, G. T. (2011). Islet amyloid polypeptide, islet amyloid, and diabetes mellitus. Physiological Reviews, 91(3), 795–826. doi: 10.1152/physrev.00042.2009
3. Raimundo, A. F., Ferreira, S., Martins, I. C., & Menezes, R. (2020). Islet Amyloid Polypeptide: A Partner in Crime With Aβ in the Pathology of Alzheimer’s Disease. Frontiers in Molecular Neuroscience, 13, 35. doi: 10.3389/fnmol.2020.00035
4. Huang, C., Lin, C., Haataja, L., Gurlo, T., Butler, A. E., Rizza, R. A., & Butler, P. C. (2007). High expression rates of human islet amyloid polypeptide induce endoplasmic reticulum stress mediated beta-cell apoptosis, a characteristic of humans with type 2 but not type 1 diabetes. Diabetes, 56(8), 2016–2027. doi: 10.2337/db07-0197
5. Abedini, A., & Schmidt, A. M. (2013). Mechanisms of islet amyloidosis toxicity in type 2 diabetes. FEBS Letters, 587(8), 1119–1127. doi: 10.1016/j.febslet.2013.01.017
6. Anguiano, M., Nowak, R. J., & Lansbury, P. T. J. (2002). Protofibrillar islet amyloid polypeptide permeabilizes synthetic vesicles by a pore-like mechanism that may be relevant to type II diabetes. Biochemistry, 41(38), 11338–11343. doi: 10.1021/bi020314u
7. Rivera, J. F., Costes, S., Gurlo, T., Glabe, C. G., & Butler, P. C. (2014). Autophagy defends pancreatic β cells from Human islet amyloid polypeptide-induced toxicity. Journal of Clinical Investigation, 124(8), 3489–3500. doi: 10.1172/JCI71981
8. Mattson, M. P., & Chan, S. L. (2003). Calcium orchestrates apoptosis. Nature Cell Biology, 5(12), 1041–1043. doi: 10.1038/ncb1203-1041
9. Tanaka, Y., Engelender, S., Igarashi, S., Rao, R. K., Wanner, T., Tanzi, R. E., Sawa, A., L. Dawson, V., Dawson, T. M., & Ross, C. A. (2001). Inducible expression of mutant α-synuclein decreases proteasome activity and increases sensitivity to mitochondria-dependent apoptosis. Human Molecular Genetics, 10(9), 919–926. doi: 10.1093/hmg/10.9.919
10. Casas, S, Novials, A., Reimann, F., Gomis, R., & Gribble, F. M. (2008). Calcium elevation in mouse pancreatic beta cells evoked by extracellular human islet amyloid polypeptide involves activation of the mechanosensitive ion channel TRPV4. Diabetologia, 51(12), 2252–2262. doi: 10.1007/s00125-008-1111-z
11. Casas, Silvia, Gomis, R., Gribble, F. M., Altirriba, J., Knuutila, S., & Novials, A. (2007). Impairment of the ubiquitin-proteasome pathway is a downstream endoplasmic reticulum stress response induced by extracellular human islet amyloid polypeptide and contributes to pancreatic beta-cell apoptosis. Diabetes, 56(9), 2284–2294. doi: 10.2337/db07-017
12. Klec, C., Ziomek, G., Pichler, M., Malli, R., & Graier, W. F. (2019). Calcium Signaling in ß-cell Physiology and Pathology: A Revisit. International Journal of Molecular Sciences, 20(24). doi: 10.3390/ijms20246110
13. Sabatini, P. V, Speckmann, T., & Lynn, F. C. (2019). Friend and foe: β-cell Ca2+ signaling and the development of diabetes. Molecular Metabolism, 21, 1–12. doi: 10.1016/j.molmet.2018.12.007
14. Aragão, D. M. O., Guarize, L., Lanini, J., da Costa, J. C., Garcia, R. M. G., & Scio, E. (2010). Hypoglycemic effects of Cecropia pachystachya in normal and alloxan-induced diabetic rats. Journal of Ethnopharmacology, 128(3), 629–633. doi: 10.1016/j.jep.2010.01.008
15. Pereira, E. D. de M., da Silva, J., Carvalho, P. da S., Grivicich, I., Picada, J. N., Salgado Júnior, I. B., Vasques, G. J., Pereira, M. A. da S., Reginatto, F. H., & Ferraz, A. de B. F. (2020). In vivo and in vitro toxicological evaluations of aqueous extract from Cecropia pachystachya leaves. Journal of Toxicology and Environmental Health. Part A, 83(19–20), 659–671. doi: 10.1080/15287394.2020.1811817
16. Campos, M. L. de, Castro, M. B. de, Campos, A. D., Fernandes, M. F., Conegundes, J. L. M., Rodrigues, M. N., Mügge, F. L. B., Silva, A. M. da, Sabarense, C. M., Castañon, M. C. M. N., Andreazzi, A. E., & Scio, E. (2021). Antiobesity, hepatoprotective and anti-hyperglycemic effects of a pharmaceutical formulation containing Cecropia pachystachya Trécul in mice fed with a hypercaloric diet. Journal of Ethnopharmacology, 280, 114418. doi: 10.1016/j.jep.2021.114418
17. Rivera-Mondragón, A., Ortíz, O. O., Bijttebier, S., Vlietinck, A., Apers, S., Pieters, L., & Caballero-George, C. (2017). Selection of chemical markers for the quality control of medicinal plants of the genus Cecropia. Pharmaceutical Biology, 55(1), 1500–1512. doi: 10.1080/13880209.2017.1307421
18. Pacheco, N. R., Pinto, N. de C. C., da Silva, J. M., Mendes, R. de F., da Costa, J. de C., Aragão, D. M. de O., Castañon, M. C. M. N., & Scio, E. (2014). Cecropia pachystachya: a species with expressive in vivo topical anti-inflammatory and in vitro antioxidant effects. BioMed Research International, 2014, 301294. doi: 10.1155/2014/301294
19. Aragão, D. M. de O., Lima, I. V. de A., da Silva, J. M., Bellozi, P. M. Q., da Costa, J. de C., Cardoso, G. M. M., de Souza-Fagundes, E. M., & Scio, E. (2013). Anti-inflammatory, antinociceptive and cytotoxic effects of the methanol extract of Cecropia pachystachya Trécul. Phytotherapy Research : PTR, 27(6), 926–930. doi: 10.1002/ptr.4811
20. Gazal, M., Kaufmann, F. N., Acosta, B. A., Oliveira, P. S., Valente, M. R., Ortmann, C. F., Sturbelle, R., Lencina, C. L., Stefanello, F. M., Kaster, M. P., Reginatto, F. H., & Ghisleni, G. (2015). Preventive Effect of Cecropia pachystachya Against Ketamine-Induced Manic Behavior and Oxidative Stress in Rats. Neurochemical Research, 40(7), 1421–1430. doi: 10.1007/s11064-015-1610-5
21. Ortmann, C. F., Réus, G. Z., Ignácio, Z. M., Abelaira, H. M., Titus, S. E., de Carvalho, P., Arent, C. O., Dos Santos, M. A. B., Matias, B. I., Martins, M. M., de Campos, A. M., Petronilho, F., Teixeira, L. J., Morais, M. O. S., Streck, E. L., Quevedo, J., & Reginatto, F. H. (2016). Enriched Flavonoid Fraction from Cecropia pachystachya Trécul Leaves Exerts Antidepressant-like Behavior and Protects Brain Against Oxidative Stress in Rats Subjected to Chronic Mild Stress. Neurotoxicity Research, 29(4), 469–483. doi: 10.1007/s12640-016-9596-6
22. Gazal, M., Ortmann, C. F., Martins, F. A., Streck, E. L., Quevedo, J., de Campos, A. M., Stefanello, F. M., Kaster, M. P., Ghisleni, G., Reginatto, F. H., & Lencina, C. L. (2014). Antidepressant-like effects of aqueous extract from Cecropia pachystachya leaves in a mouse model of chronic unpredictable stress. Brain Research Bulletin, 108, 10–17. doi: 10.1016/j.brainresbull.2014.07.007
23. Raimundo, A. F., Ferreira, S., Farrim, M. I., Santos, C. N., & Menezes, R. (2020). Heterologous Expression of Immature Forms of Human Islet Amyloid Polypeptide in Yeast Triggers Intracellular Aggregation and Cytotoxicity. Frontiers in Microbiology, 11, 2035. doi: 10.3389/fmicb.2020.02035
24. Costa, G. M., Ortmann, C. F., Schenkel, E. P., & Reginatto, F. H. (2011). An HPLC‐DAD method to quantification of main phenolic compounds from leaves of Cecropia species. Journal of the Brazilian Chemical Society, 22, 1096–1002. doi: 10.1590/S0103-50532011000600014
25. Gietz, R. D., & Schiestl, R. H. (1991). Applications of high efficiency lithium acetate transformation of intact yeast cells using single-stranded nucleic acids as carrier. Yeast, 7(3), 253–263. doi: 10.1002/yea.320070307
26. Garcia, G., Santos, C. N. do, & Menezes, R. (2016). High-Throughput Yeast-Based Reporter Assay to Identify Compounds with Anti-inflammatory Potential. Methods in Molecular Biology (Clifton, N.J.), 1449, 441–452. doi: 10.1007/978-1-4939-3756-1_29
27. Araki, Y., Wu, H., Kitagaki, H., Akao, T., Takagi, H., & Shimoi, H. (2009). Ethanol stress stimulates the Ca2+-mediated calcineurin/Crz1 pathway in Saccharomyces cerevisiae. Journal of Bioscience and Bioengineering, 107(1), 1–6. doi: 10.1016/j.jbiosc.2008.09.005
28. Dudnik, A., Almeida, A. F., Andrade, R., Avila, B., Bañados, P., Barbay, D., Bassard, J.-E., Benkoulouche, M., Bott, M., Braga, A., Breitel, D., Brennan, R., Bulteau, L., Chanforan, C., Costa, I., Costa, R. S., Doostmohammadi, M., Faria, N., Feng, C., … Forster, J. (2018). BacHBerry: BACterial Hosts for production of Bioactive phenolics from bERRY fruits. Phytochemistry Reviews, 17(2), 291–326. doi: 10.1007/s11101-017-9532-2
29. Menezes, R., Foito, A., Jardim, C., Costa, I., Garcia, G., Rosado-Ramos, R., Freitag, S., Alexander, C. J., Outeiro, T. F., Stewart, D., & Santos, C. N. (2020). Bioprospection of Natural Sources of Polyphenols with Therapeutic Potential for Redox-Related Diseases. Antioxidants, 9(9). doi: 10.3390/antiox9090789
30. Kallscheuer, N., Menezes, R., Foito, A., da Silva, M. H., Braga, A., Dekker, W., Sevillano, D. M., Rosado-Ramos, R., Jardim, C., Oliveira, J., Ferreira, P., Rocha, I., Silva, A. R., Sousa, M., Allwood, J. W., Bott, M., Faria, N., Stewart, D., Ottens, M., … Marienhagen, J. (2019). Identification and Microbial Production of the Raspberry Phenol Salidroside that Is Active against Huntington’s Disease. Plant Physiology, 179(3), 969–985. doi: 10.1104/pp.18.01074
31. Cheng, B., Liu, X., Gong, H., Huang, L., Chen, H., Zhang, X., Li, C., Yang, M., Ma, B., Jiao, L., Zheng, L., & Huang, K. (2011). Coffee Components Inhibit Amyloid Formation of Human Islet Amyloid Polypeptide in Vitro: Possible Link between Coffee Consumption and Diabetes Mellitus. Journal of Agricultural and Food Chemistry, 59(24), 13147–13155. doi: 10.1021/jf201702h
32. Karthikesan, K., Pari, L., & Menon, V. P. (2010a). Combined treatment of tetrahydrocurcumin and chlorogenic acid exerts potential antihyperglycemic effect on streptozotocin-nicotinamide-induced diabetic rats. General Physiology and Biophysics, 29(1), 23—30. doi: 10.4149/gpb_2010_01_23
33. Karthikesan, K., Pari, L., & Menon, V. P. (2010b). Protective effect of tetrahydrocurcumin and chlorogenic acid against streptozotocin–nicotinamide generated oxidative stress induced diabetes. Journal of Functional Foods, 2(2), 134–142. doi: 10.1016/j.jff.2010.04.00
34. Miao M, & Xiang L. (2020). Pharmacological action and potential targets of chlorogenic acid. Advances in Pharmacology, 87, 71–88. doi: 10.1016/bs.apha.2019.12.002
35. Kolb, H., Martin, S., & Kempf, K. (2021). Coffee and Lower Risk of Type 2 Diabetes: Arguments for a Causal Relationship. Nutrients, 13(4). doi: 10.3390/nu13041144
36. Ziqubu, K., Dludla, P. V, Joubert, E., Muller, C. J. F., Louw, J., Tiano, L., Nkambule, B. B., Kappo, A. P., & Mazibuko-Mbeje, S. E. (2020). Isoorientin: A dietary flavone with the potential to ameliorate diverse metabolic complications. Pharmacological Research, 158, 104867. doi: 10.1016/j.phrs.2020.104867
37. Gilon, P., Chae, H.-Y., Rutter, G. A., & Ravier, M. A. (2014). Calcium signaling in pancreatic β-cells in health and in Type 2 diabetes. Cell Calcium, 56(5), 340–361. doi: 10.1016/j.ceca.2014.09.001
38. Jung, H.-J., Im, S.-S., Song, D.-K., & Bae, J.-H. (2017). Effects of chlorogenic acid on intracellular calcium regulation in lysophosphatidylcholine-treated endothelial cells. BMB Reports, 50(6), 323–328. doi: 10.5483/bmbrep.2017.50.6.182
