![]()
Biomedical Sciences, Biomed Biopharm Res., 2021; 18(2):200-212
doi: 10.19277/bbr.18.2.269; PDF version [+] here; Portuguese html [+] here
Optoaccoustic Tomography – good news for microcirculatory research
Tiago Granja, Sérgio Faloni de Andrade, and Luis Monteiro Rodrigues *
CBIOS—Universidade Lusófona’s Research Center for Biosciences & Health Technologies, Campo Grande 376, 1749-024 Lisboa, Portugal
*corresponding author:
Abstract
Tomographic imaging is a well established technology in preventive medicine and biomedical research, although not without limitations and concerns. Optoacoustic tomography (OAT) is a recent development that bridges optical and sonographic techniques to solve spatial resolution in deep-tissue imaging. In addition to safety advantages, OAT allows multiple wavelength readings for natural thermoelastic chromophores. In this study, we explore Multi Spectral Optoacoustic Tomography (MSOT) capacities to simultaneously acquire three independent chromophores – deoxygenated haemoglobin (Hb), oxygenated haemoglobin (HbO2), and melanin, from healthy human volunteers, with maximal molar extinction of HbO2 at 950 nm, Hb at 750 nm and melanin at 680 nm. Later we demonstrate how image stability during acquisition is fundamental for optimal resolution, precision and consistency of high throughout MSOT data collection. From recorded scans, a workflow is layered for data evaluation. With the MSOT dedicated software results were extracted from 3D image analysis of deep (15 mm3) vessels. The possibilities offered by this new system, specially in vascular pathophysiology, are immense and can be extended beyond current knowledge.
Keywords: Optoacoustic tomography; MSOT; haemoglobin; microcirculation; vascular research
Received: 17/09/2021; Accepted: 30/12/2021
Introduction
The importance of tomography in preventive medicine as in biomedical research is well established. Tomographic techniques such as computed tomography (CT), single-photon emission computed tomography (SPECT), and positron emission tomography (PET) offer different image resolution and contrast, meaning also different applicabilities, costs, and limitations. (1-4). A major limitation of this technology is spatial resolution in deep-tissue imaging due to light diffusion effects (4,5). Recent advances in photonics and optical imaging have drawn our attention to new optical tomography systems such as diffuse optical tomography (DOT), fluorescence molecular tomography (FMT), bioluminescence tomography (BLT), and photoacoustic tomography (PAT). These new developments aim to improve sensitivity and precision, and include new learning methods to provide more accurate and reliable reconstruction analysis (5,6).
Photoacoustic tomography (PAT) is a particularly interesting imaging technology, as it combines optics and ultrasound principles rather than classical ionizing radiation. It is also known as thermoacoustic tomography (TAT) or, more currently, optoacoustic tomography (OAT). This technology uses laser light of different frequencies to heat and expand the targeted tissue, creating a sonographic wave that can be measure at skin surface (6-9). This signal can then be used to produce an image by specific reconstruction algorithms. Different biological components can be targeted by different laser wavelengths. Compared with similar technologies, several advantages of this technique can be identified. PAT does not use ionizing radiation, in contrast to classical CT PET and or MRI, and is faster, easier, and less expensive to operate. Compared with DOT or with FMT, PAT shows better penetration capacities with high resolution, showing better contrast and fewer artifacts than sonography (6-10), the most common technology for cardiovascular diagnostics (9,10).
PAT applications have expanded significantly in the last years, from basic investigation to clinical applications, also in cardiovascular medicine (10-13). A recent development of this technology is the use of lasers with multiple wavelengths – the multi-spectral optoacoustic tomography (MSOT) – which allows the (in vivo) identification of several chromophores without contrast agents. These chromophores might be regarded as endogenous biomarkers of microcirculatory function, such as oxygenated haemoglobin (HbO2), deoxygenated haemoglobin (Hb), or the mean saturation of oxygen (mSO2) (14-18). Other chromophores, including lipids, melanin and water might also be detected and visualised for functional analysis (10,16). In the present study we explore the operation and capabilities of MSOT in a set of healthy human participants.
Materials and Methods
Participants
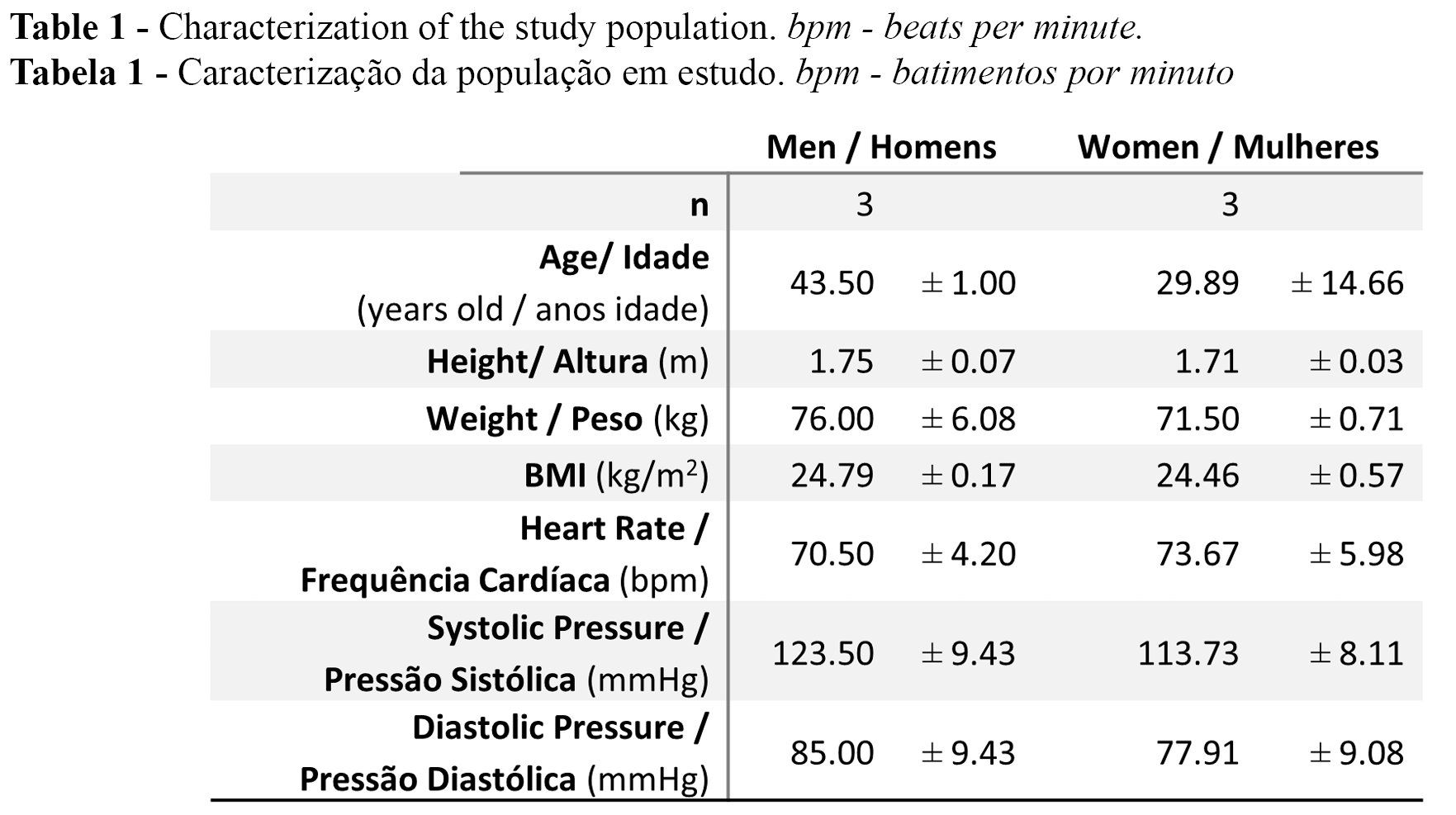
Our convenience sample included six healthy participants of both genders (3 men and 3 women), 18 to 60 years old (mean + sd 36.70 ± 15.16), selected following specific inclusion / non-inclusion criteria. Participants were normotensive, nonsmokers, and free of any medication or food supplementation. Restrictions included caffeine and alcohol consumption 24 hours prior to measurements, and the use any topical formulations (including cosmetic) on the skin. Participants reported regular physical activity and absence of any metabolic disease (diabetes, dyslipidaemia). Table 1 summarises the full characterization of the participants. All study procedures followed the principles established by the Helsinki Declaration and subsequent amendments (19) and were previously approved by the Institutional Ethics Committee.
Multi Spectra Optoacoustic Tomography and Image Analysis
A MSOT optoacoustic imaging system from Acuity (iThera Medical GmbH, Munich, Germany) was used for all analyses. This instrument is composed of a Nd:YAG laser, which produces excitation pulses of 9 ns at wavelengths from 680 nm to 980 nm. The MSOT system repetition rate can reach 50 Hz with a wavelength tuning speed of 10 ms and a peak pulse energy of 35 mJ at 730 nm. A fiber-optic bundle integrated into the detector produces a circular spot of light of approximately 10 mm diameter. In this system, ultrasound detection is made by 256 ultrasound transducers with a centre frequency of 8 MHz (60% bandwidth), organized in a hemispherical array of 98° angular coverage and a radius of curvature of 30 mm. The laser light is conducted to a 3D probe by an optical fiber of 15 mm3. After scanning, images were processed for fluence correction, optical focusing given by the speed of sound and optoacoustic thermoelastic signals of the selected chromophores for Hb and HbO2 were reconstructed, and Regions of Interest (ROIs) were analysed with the iThera Medical ViewMSOT software.
ImageJ software (National Institutes of Health, USA v.1.53k14 from 26 June 2021) was used to analyse the merged, (re)constructed, image from individual images of X, Y and Z axis scans.
Experimental Procedure
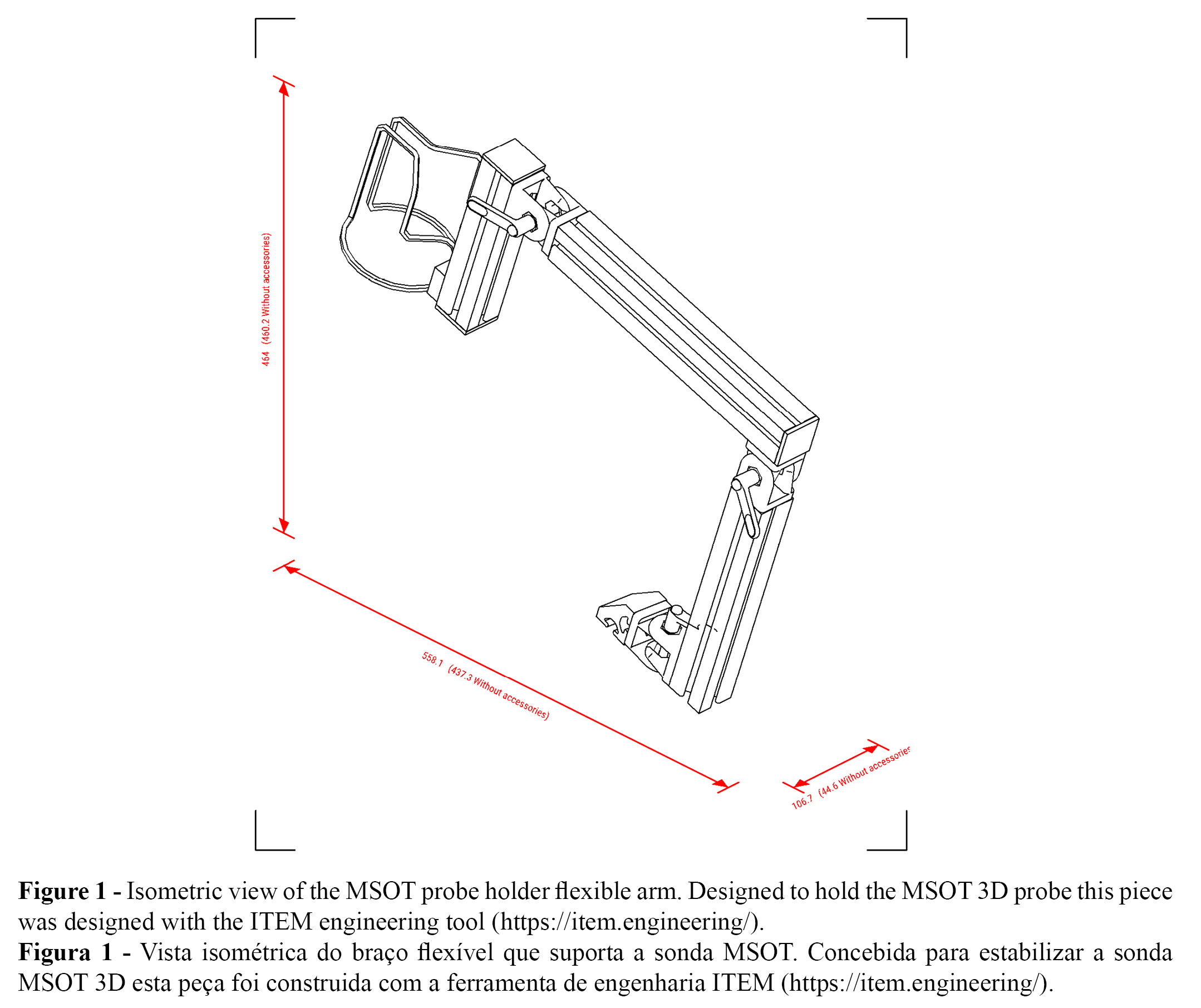
The ventral aspect of a randomly chosen forearm, with body hair chemically removed immediately prior to analysis, was marked for probe application (4,20). The MSOT 3D probe was then applied and focused in a fixed orientation. A thin layer of gel for sonography was applied in the MSOT 3D cup before contact with the skin to improve image acquisition conditions. The MSOT 3D cup was locked in position with a self-assembled arm (ITEM – Engineering,Solingen ,Germany)(Figure 1).
Statistics
Statistical analysis was performed with GraphPad Prism 9.2.0, MachineID: 0861F12DB8D10, and normal distribution was tested with the Shapiro-Wilk test and direct observation of QQ plots. Except where noted, all data is presented as mean ± sd. Significance was observed through t test of selected comparisons, analysed with 95% confidence level.
Results and Discussion
The MSOT detects numerous chromophore signals at a repetition rate of 50 Hz with a wavelength tuning speed of 10 ms. MSOT wavelengths range from near visible to infra-red imaging (680 to 980 nm), allowing non-invasive analysis up to 15 mm deep and producing regions of analysis of 15 mm3 from the recording of 10 frames (in 2 seconds) for each (X, Y and Z) axis for every (preset) thermoelastic chromophore. Micrometre resolution of selected regions yield HbO2, Hb, and melanin data from 3D reconstructed images (using a single-element ultrasonic transducer and a curved ultrasound 2-D sensor), referred as the most important biomarkers from OAT (4,15,18,21,22).
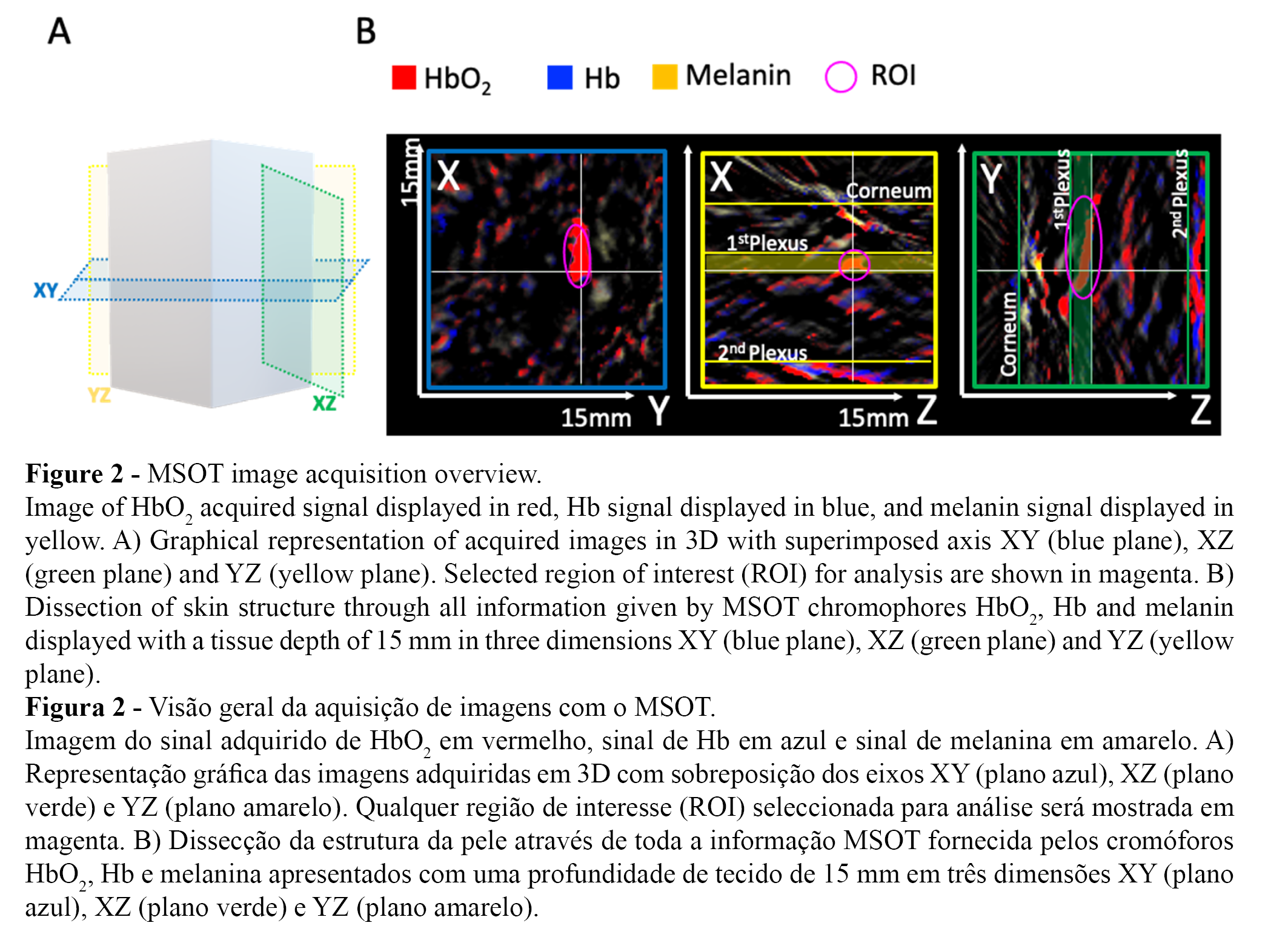
The MSOT can virtually be instructed to ”unmix” up to three different thermoelastic chromophores (certified by the manufacturer) and loaded as pre-sets before any acquisition. As discussed, for our study, the instrument was set up to observe signals from HbO2, Hb, and melanin. While acquiring from any given region (e.g., the skin), an image stack composed of XY, XZ, and YZ axes is generated (Figure 2A). At a depth of 15 mm, the images produced have a resolution of 150 µm, enabling the identification of the first and second vascular plexuses of the skin.
Spectra unmixing and image merge
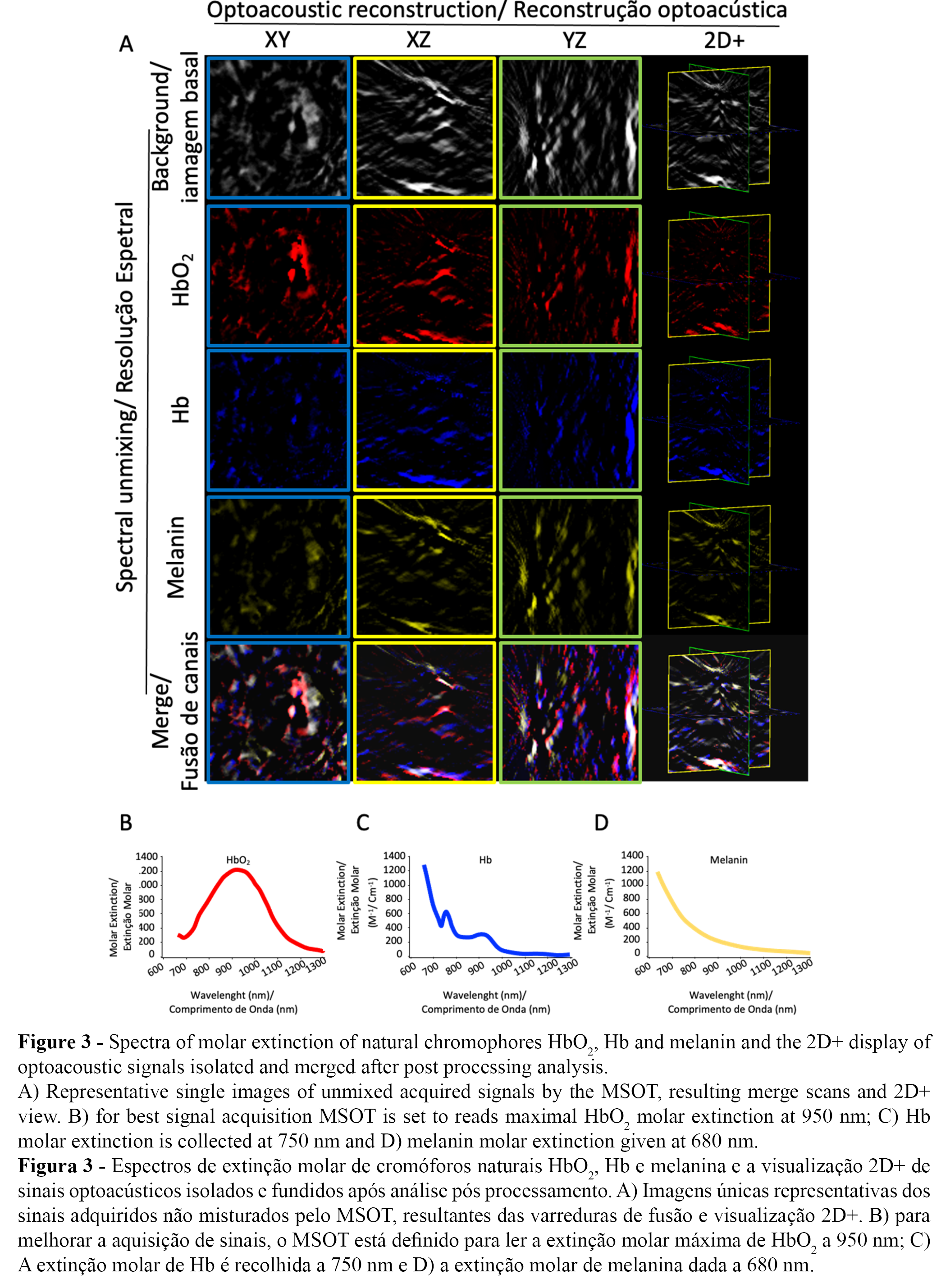
Objects generated during acquisition are three dimensional (3D), and the screenshot view allows measurements and annotations as shown in Figure 2B. To extract data from acquired images, prior analysis requires assembly of multiple thermoelastic signals and post processing by the iThera MSOTview software. This process follows a reconstruction sequence of optoacoustic signals - the correction of the speed of sound for calibration of best focus, application of fluence correction, and calculation of spectral unmixing for the acquired signals of HbO2, Hb and melanin (Figure 3A). For best signal acquisition, the MSOT reads maximal HbO2 molar extinction at 950 nm, Hb at 750nm, and melanin at 680nm (Figure 3B-D). Representative images of each of the acquired signals of HbO2, Hb, and melanin demonstrate the perfect unmixing of the acquired signals in all XY, XZ, and YZ axes and the plot of the respective merged image (Figure 3A). Additionally, a 2D+ image view is generated for optimal navigation over the 3D images acquired and to adjust proper colour intensities. Single chromophore images are fundamental to set correct colour lookup tables and to enable independent study of the acquired signals.
Optoaccoustic reconstruction and Region of Interest (ROI) analysis
After image reconstruction, fluence correction, focusing of speed of sound, unmixing, and adjustment of color balance and saturation of preferred lookup tables, each chromophore signal acquired by the MSOT can be analysed. To overcome signal flattening from signal-to-noise ratio derived from large image analysis, elements of each 3D scan must be selected as Regions-Of-Interest (ROI) before data analysis. ROI analysis (depicted in Figure 4A) follows a microvasculature ROI structure in 9 of all 10 frames acquired. Each frame is of 150 µm of tissue depth in X, Y and Z axes. To acquire data from any ROI, each frame must follow the selected ROI across XZ (yellow bar in depicted fames) and YZ (green bar in depicted frames) while a polygon selection tool (magenta selected area) delineates the ROI in XY (blue frames). On the 2D+ image overview, the 3D area of analysis is built (Figure 4A).
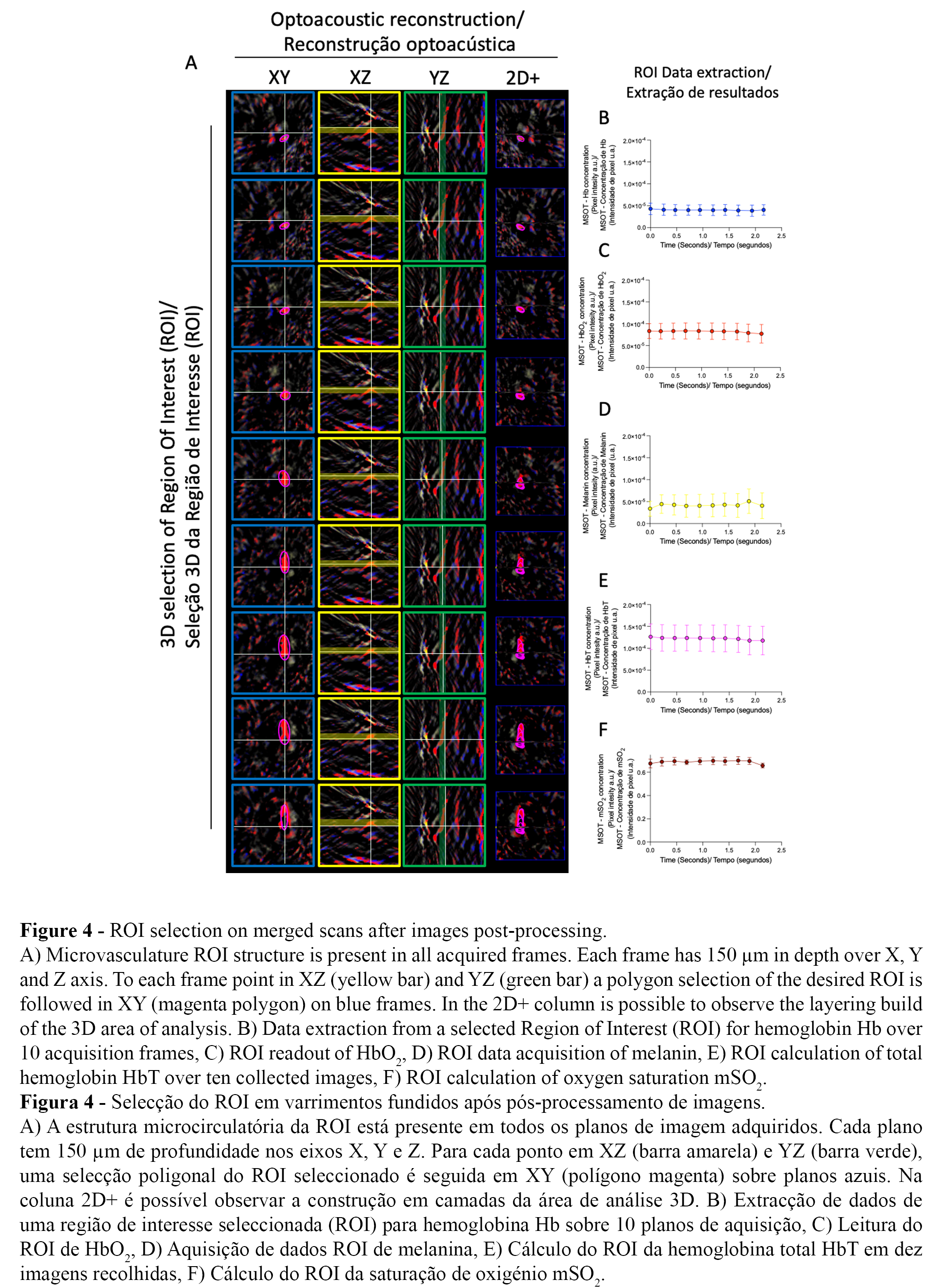
Biomarkers
Through the MSOTview each scanned ROI exported data is provided in bursts of ten images derived from optoacoustic reconstruction. ROI selection of MSOT acquired images allows calculation of ROI volume (mm3), as well as the concentrations of HbO2 (in arbitrary units a.u.), oxidized Hb (a.u.) and melanin (a.u.) (4,14,18,20).
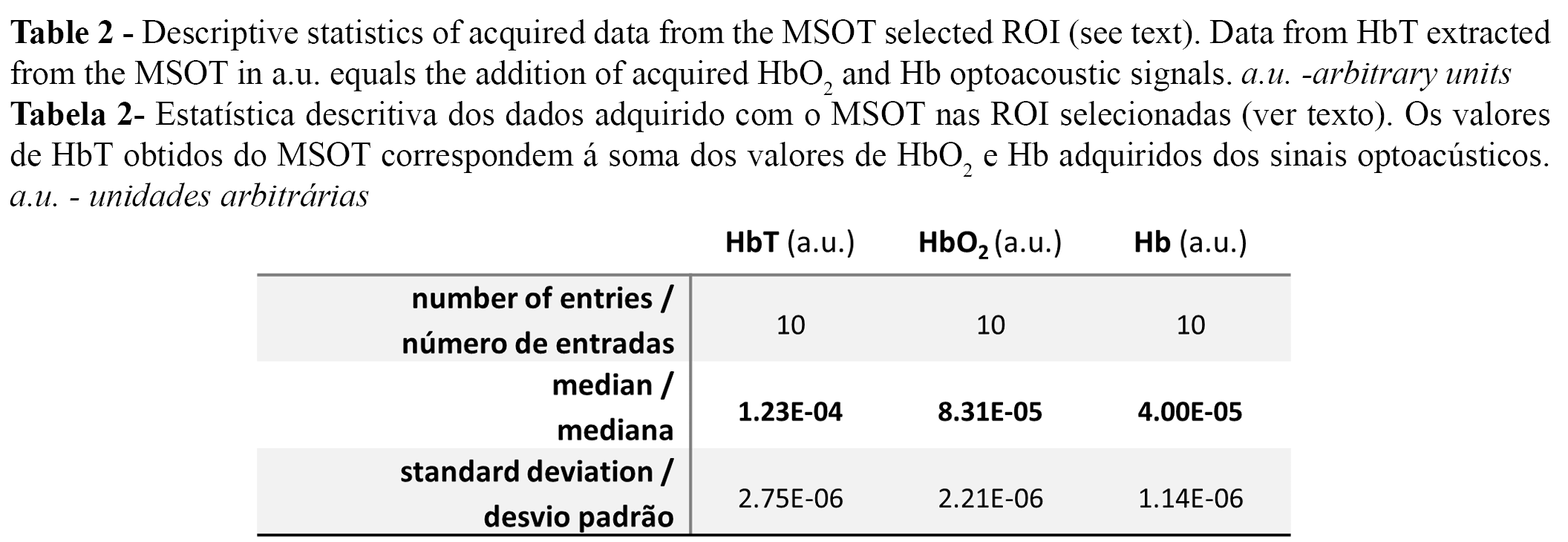
Overall, the acquisition speed of ten images of three chromophores over a reconstruction of sixse wavelengths (700 nm, 730 nm, 760 nm, 800 nm, 850 nm, and 900 nm) is constant at 2s. The data stability from the selected analysed ROI is depicted by the linearity of the signal recorded for HbO2, deoxygenated Hb, and melanin (a.u.) (Figure 4B-D). From HbO2 and Hb signals, the MSOTview is able to calculate the total haemoglobin (HbT) and the mean oxygen saturation (mSO2) (Figure 4E-F) (14).
The MSOT calculated HbT (1.23E-04±2.75E-06 a.u) is therefore given by the addition Hb (of 4.00E-05±1.14E-06) and HbO2 (of 8.31E-05±2.21E-06) signal intensities shown in Table 2. Data evaluation plotted in Figure 5A summarizes the scans taken from our six volunteers over a total of ten optoacoustic images in 2s. Data from Hb (line in blue) and HbO2 (line in red) consistently match consistently their addition as HbT (line in pink) over the course of the optoacoustic acquisitions. We also observe that in resting position, HbO2 concentration is statistically higher than Hb in all volunteers (Figure 5B), and that value of mSO2 is consistently above the 0.7 a.u. oxygen saturation signal input cut-off (Figure 5C).
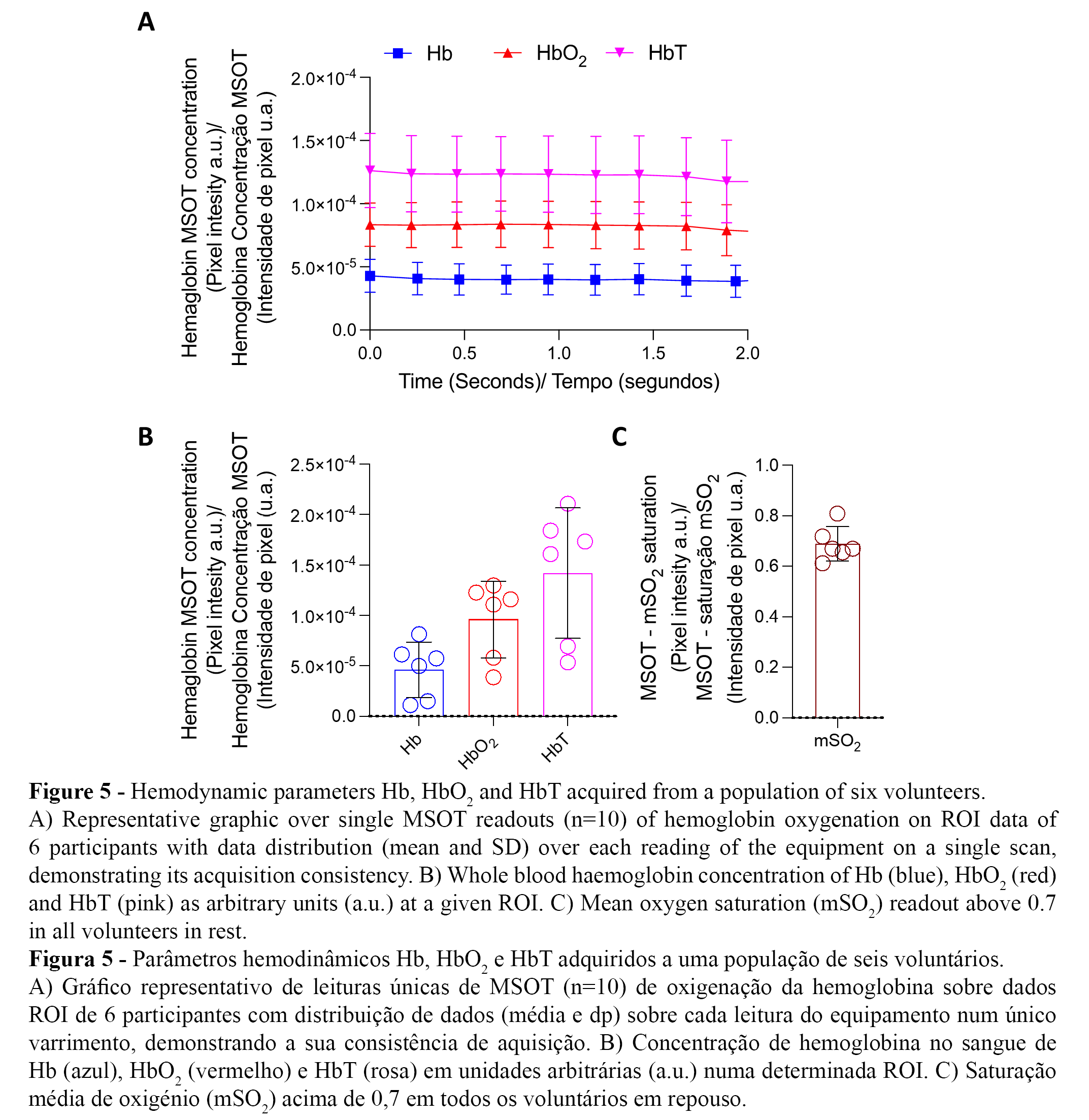
Our approach is in-line with previously published studies showing that that image acquisition with the MSOT system is extremely intuitive (even in 3D mode). Discrimination of the skin microvasculature stratification into upper and lower skin plexus is visualized on demand in different axis (X, Y, Z) at a volume of 15 mm3 (4,14-16). The application of ROI analysis shows high descriptive potential as it enables the MSOT to independently and simultaneously evaluate the blood volume and oxygenation of the different skin plexus with different / multiple selected ROI’s. Additionally, MSOT selected pre-set offers the possibility to acquire and unmix (at least) three independent chromophores of interest (Hb, HbO2, and melanin in our study) simultaneously.
In any case, the complete stabilization of the 3D probe during image acquisition is critical for the acquisition quality and for data analysis, such that the use of a fixed stand to secure the probe is essential.
We recognize that the data processing involved is complex, and requires an experienced operator and analyst. However, the possibilities offered by this new system to bring new perspectives especially to vascular pathophysiology research are immense and can be extended beyond current knowledge.
Authors Contributions Statement
LMR, TG conceptualization and study design; TG, SA experimental implementation; TG, data analysis and data curation; TG, LMR, drafting and reviewing; LMR, supervision and final writing.
Funding
This research is funded by ALIES and COFAC principal providers of the technology under study, and by Fundação para a Ciência e a Tecnologia (FCT) through the grant UIDB/04567/2020 to CBIOS.
Conflict of Interests
Editors involved in this manuscripts’ authorship had no participation in the review or decision process. All authors have stated that there are no financial and/or personal relationships that could represent a potential conflict of interest.
Acknowledgements
The authors would like to express their thanks to CBIOS staff for their effort supplying all the necessary help, and to all the participants involved in the study.
References
1. Yodh, A., Chance B. (1995). Spectroscopy and imaging with diffusing light Physics Today 48, 34–40. doi.org/10.1063/1.881445
2. Ntziachristos, V., Ripoll, J., Wang, L. V., & Weissleder, R. (2005). Looking and listening to light: the evolution of whole-body photonic imaging. Nature biotechnology, 23(3), 313–320. doi.org/10.1038/nbt1074
3. Rubin G. D. (2014). Computed tomography: revolutionizing the practice of medicine for 40 years. Radiology, 273(2 Suppl), S45–S74. doi.org/10.1148/radiol.14141356
4. Karlas, A., Fasoula, N. A., Paul-Yuan, K., Reber, J., Kallmayer, M., Bozhko, D., Seeger, M., Eckstein, H. H., Wildgruber, M., & Ntziachristos, V. (2019). Cardiovascular optoacoustics: From mice to men - A review. Photoacoustics, 14, 19–30. doi.org/10.1016/j.pacs.2019.03.001
5 Tan, H., Gu, Y., Yu, H., Hu, P., Zhang, Y., Mao, W., & Shi, H. (2020). Total-Body PET/CT: Current Applications and Future Perspectives. AJR. American journal of roentgenology, 215(2), 325–337. doi.org/10.2214/AJR.19.22705
6. Zhang L, Zhang G. (2019) Brief review on learning-based methods for optical tomography. Journal of Innovative Optical Health Sciences, 12, 1930011. doi: 10.1142/S17935458193001185
7. Hu, S., & Wang, L. V. (2010). Photoacoustic imaging and characterization of the microvasculature. Journal of biomedical optics, 15(1), 011101. doi.org/10.1117/1.3281673
8. Zhang, H. F., Maslov, K., Stoica, G., & Wang, L. V. (2006). Functional photoacoustic microscopy for high-resolution and noninvasive in vivo imaging. Nature biotechnology, 24(7), 848–851. doi.org/10.1038/nbt1220
9. Merčep, E., Jeng, G., Morscher, S., Li, P. C., & Razansky, D. (2015). Hybrid optoacoustic tomography and pulse-echo ultrasonography using concave arrays. IEEE transactions on ultrasonics, ferroelectrics, and frequency control, 62(9), 1651–1661. doi.org/10.1109/TUFFC.2015.007058
10. Wang L. V. (2009). Multiscale photoacoustic microscopy and computed tomography. Nature photonics, 3(9), 503–509. doi.org/10.1038/nphoton.2009.157
11. Haedicke, K., Agemy, L., Omar, M., Berezhnoi, A., Roberts, S., Longo-Machado, C., Skubal, M., Nagar, K., Hsu, H. T., Kim, K., Reiner, T., Coleman, J., Ntziachristos, V., Scherz, A., & Grimm, J. (2020). High-resolution optoacoustic imaging of tissue responses to vascular-targeted therapies. Nature biomedical engineering, 4(3), 286–297. doi.org/10.1038/s41551-020-0527-8
12. Nitkunanantharajah, S., Haedicke, K., Moore, T. B., Manning, J. B., Dinsdale, G., Berks, M., Taylor, C., Dickinson, M. R., Jüstel, D., Ntziachristos, V., Herrick, A. L., & Murray, A. K. (2020). Three-dimensional optoacoustic imaging of nailfold capillaries in systemic sclerosis and its potential for disease differentiation using deep learning. Scientific reports, 10(1), 16444. https://doi.org/10.1038/s41598-020-73319-2.
13. Haedicke, K., Brand, C., Omar, M., Ntziachristos, V., Reiner, T., & Grimm, J. (2017). Sonophore labeled RGD: a targeted contrast agent for optoacoustic imaging. Photoacoustics, 6, 1–8. doi.org/10.1016/j.pacs.2017.03.001.
14. Zafar, H., Breathnach, A., Subhash, H. M., & Leahy, M. J. (2015). Linear-array-based photoacoustic imaging of human microcirculation with a range of high frequency transducer probes. Journal of biomedical optics, 20(5), 051021. doi.org/10.1117/1.JBO.20.5.051021
15. Yao, J., Xia, J., & Wang, L. V. (2016). Multiscale Functional and Molecular Photoacoustic Tomography. Ultrasonic imaging, 38(1), 44–62. doi.org/10.1177/0161734615584312
16. Gujrati, V., Mishra, A., & Ntziachristos, V. (2017). Molecular imaging probes for multi-spectral optoacoustic tomography. Chemical communications (Cambridge, England), 53(34), 4653–4672. https://doi.org/10.1039/c6cc09421j
17. Luís Dean-Ben, X., & Razansky, D. (2018). Localization optoacoustic tomography. Light, science & applications, 7, 18004. doi.org/10.1038/lsa.2018.4
18. Karlas, A., Masthoff, M., Kallmayer, M., Helfen, A., Bariotakis, M., Fasoula, N. A., Schäfers, M., Seidensticker, M., Eckstein, H. H., Ntziachristos, V., & Wildgruber, M. (2021). Multispectral optoacoustic tomography of peripheral arterial disease based on muscle hemoglobin gradients-a pilot clinical study. Annals of translational medicine, 9(1), 36. doi.org/10.21037/atm-20-3321
19. World Medical Association. (2013) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310(20):2191–2194. doi:10.1001/jama.2013.281053
20. Taruttis, A., Timmermans, A. C., Wouters, P. C., Kacprowicz, M., van Dam, G. M., & Ntziachristos, V. (2016). Optoacoustic Imaging of Human Vasculature: Feasibility by Using a Handheld Probe. Radiology, 281(1), 256–263. doi.org/10.1148/radiol.2016152160
21. Kneipp, M., Turner, J., Hambauer, S., Krieg, S. M., Lehmberg, J., Lindauer, U., & Razansky, D. (2014). Functional real-time optoacoustic imaging of middle cerebral artery occlusion in mice. PloS one, 9(4), e96118. doi.org/10.1371/journal.pone.0096118
22. van den Berg, P. J., Daoudi, K., & Steenbergen, W. (2015). Review of photoacoustic flow imaging: its current state and its promises. Photoacoustics, 3(3), 89–99. doi.org/10.1016/j.pacs.2015.08.001
