![]()
Biopharmaceutical Sciences, Biomed Biopharm Res., 2021; 18(2): 263-273
doi: 10.19277/bbr.18.2.273; [+] PDF version here; [+] Portuguese html version here
Clinical efficacy of a cosmetic formulation with vitamin B3 for the treatment of oily skin
Marcella Gabarra Almeida Leite & Patrícia Maria Berardo Gonçalves Maia Campos*
Faculdade de Ciências Farmacêuticas de Ribeirão Preto, Universidade de São Paulo, Av. do Café S/N, Monte Alegre, 14040-903, Ribeirão Preto, SP, Brasil
*corresponding author:
Abstract
Oily skin has special needs, as the treatment and prevention of non-inflammatory acne and sebum control, maintaining skin barrier function and hydration. In this context, the importance of the development of specific and innovative cosmetics formulations to to fulfill the needs of this type of skin is evident. In this view, the aim of this study was the evaluation of the clinical efficacy of a cosmetic formulation containing Vitamin B3 in the treatment of oily skin using biophysical and skin imaging techniques. Female volunteers (n=40) between 18 to 30 years old with oily skin and/or mild inflammatory acne were recruited and received a formulation containing, or not (vehicle), 3% of Vitamin B3. Stratum corneum water content, transepidermal water loss (TEWL), sebum content and quantity of porphyrins were analyzed before (baseline) and after 28 days of application. Finally, an efficacy perception questionnaire was applied at the study end. The formulation with vitamin B3, showed a significant reduction of sebum and porphyrin content. The study participants in the assessment of perceived efficacy also reported these results. The studied formulation with Vitamin B3 was effective for oily skin care, acting on important parameters, such as controlling oiliness and improving skin appearance.
Keywords: cosmetics, Niacinamide, skin physiology, oily skin, biophysical and skin imaging
Received: 01/12/2021; Accepted: 31/12/2021
Introduction
Oily skin, also known as seborrhea, is one of the most common skin issues, most common in teenagers and young adults. In this type of skin, the sebaceous glands are characteristically oversized, leading to an excessive production of sebum, which causes a shiny and greasy appearance and can collaborate with the appearance of acne (1). It is also described as a thickening of the skin with the presence of enlarged pores, being more visible on the nose and forehead regions (2). These characteristics are usually esthetically disturbing, and are perceived as a serious cosmetic problem, with a negative influence on quality of life and self-perception (3).
This condition also requires special needs, as well as the treatment and prevention of non-inflammatory acne and control of sebum content, along with the integrity of skin barrier function and hydration. In this way, the application of vitamins in cosmetic formulations is an excellent alternative of treatment for this skin condition, and thus an important topic of interest for dermatological research.
Vitamin B3, also called Niacinamide, is described in the literature due to its many properties on the skin, including antiinflamatory, antipruritic, lightening, sebostatic and antimicrobial effects (4).
A study from Khodaeian and collaborators (2013) compared the use of vitamin B3 with clindamycin, which is extensively used in the dermatological field for the treatment of inflammatory acne and observed great results. This was also described by Shalita et al. (1995), reporting its use as a good alternative to the use of antibiotics and more advantageous due to the other reported benefits to the skin (5,6).
Nicotinamide, which is an amide form of vitamin B3, has the capacity of boosting cellular energy. It also regulates poly-ADP-ribose-polymerase 1, an enzyme with important roles in DNA repair and in the expression of inflammatory cytokines, which are useful for the treatment of a wide range of dermatological conditions, including acne (7).
In this context, vitamin B3 has potential benefits for the treatment of oily skin conditions, controlling the pathogenic factor of acne (Cutibacterium acnes) given to Niacinamide’s antimicrobial properties as well as controlling the excessive production of sebum.
Finally, considering that a previous study of our research group showed the benefits of the application of vitamin B3 in association with vitamin B6 and Zinc-PCA in the treatment of oily skin with inflammatory acne (8), the aim of the present study was to evaluate the clinical efficacy of formulations containing vitamin B3 for the treatment of oily skin using biophysical and skin imaging techniques.
Materials and Methods
Studied Formulation
Two topical formulations of gel creams based on xanthan gum, tribehenine esters PEG-20, octyldodecyl myristate, polypropylene and sorbitan PEG-10, added or not (vehicle) with 3% vitamin B3 - nicotinamide (Table 1) were prepared.
Study design
The present study was a monocentric, double-blinded, randomized, placebo-controlled study on the effects of a cosmetic formulation with added Vitamin B3 on oily skin after four weeks of treatment. The study was approved by the Ethics Committee of the Faculty of Pharmaceutical Sciences of Ribeirão Preto/SP (CEP / FCFRP 248/2012) and followed current Good Clinical Practice regulations.
All test subjects received detailed information listing every single relevant parameter to the study and all the procedures involved. All subjects gave signed informed consent after written information and a possibility for further questioning. The study was performed in accordance with the ethical standards of the Declaration of Helsinki of 1975, as revised in 2013.
Participants
A total of 40 healthy female subjects were enrolled in the study: 20 subjects were randomized to each of two treatment groups using the cosmetic formulations under study, which included or not 3% Vitamin B3 (Nicotinamide). The volunteers were instructed to apply the formulation to the face twice daily for a period of four weeks. Baseline and final measures were done in the frontal (forehead), malar (cheeks) and chin regions.
The inclusion criteria for this study were healthy females ranging in age from 18-30 years (homogeneous distribution between treatment groups); presenting oily skin and acne, phototype II-IV (Fitzpatrick scale); general good health and mental condition; avoid sun exposure during the study period; personal informed consent to participate in the study; personal presence on the predefined days at the institute, and willingness and capability to follow the study rules and a fixed schedule, know that the data could be used to share the project. The study participants were also instructed not to use other cosmetic products and to maintain their alimentary habits during the study period.
The exclusion criteria was as follows: any deviation from the above-mentioned inclusion criteria: pregnancy (or intention to become pregnant) or in period of breast feeding; skin diseases (e.g., atopic eczema, neurodermatitis or psoriasis) on the test areas or other dermatological disorders (e.g., scars, sunburn or moles), whose therapy could influence the results of the study, such as systemic steroids or antibiotics, steroids or local immunomodulatory topics three months prior to the study; smoking habit; severe disorders within 6 months prior to study start (e.g., cancer, acute cardiac and circularity disorders, severe diabetes, or alcohol or drug abuse); history of medical or surgical events that could significantly affect the outcome of the study, including any cardiovascular disease, skin disease, gastrointestinal diseases, indigestion, hypertension (>160/95 mm Hg on repeated measurements); participation in any other clinical study; medical treatments on the study area 30 days before the study start; use of tanning beds or self-tanning products a month before or during the study; any other condition that, in the opinion of the investigator, may interfere with the results or involve a risk to the subject (9).
Assessments
The test areas were frontal (forehead), malar (cheeks) and chin regions, with sides randomly chosen. Prior to every measurement, the subjects were required to expose their uncovered test areas to the indoor climate conditions (21.5 ± 1 ºC and 50 ± 5% relative humidity) for 20 min.
Measurements were made immediately before starting the product treatment (baseline values) and after 14 (t14) and 28 days (t28) of study.
The transepidermal water loss (TEWL) measurement was carried out with a Tewameter® TM 210 (Courage Khazaka, Electronic GmbH, Germany), which measures the percentage relative humidity at the skin surface. It uses a sensing electrode encased in a proper probe and is based on the principles described by Adolf Fick in 1885. The values are registered in g/m²·h. The probe of the device remained in contact with the skin to obtain 45 measurements in three study areas. Three measurements were made in each study area and the average of the obtained values was calculated (10).
The casual sebum level was determined with a photometric device (Sebumeter® SM 815, Courage+Khazaka, Cologne, Germany). A special opaque plastic tape (64 mm2) was pressed onto the skin for 30 s with a slight pressure to collect the sebum. The resulting increase in the transparency of the tape was measured, where the displayed values correspond to the sebum amount on the skin surface in micrograms of sebum per square centimeter (11).
To count porphyrins in follicular surface, a Visiopor® PP 34 system (Courage+Khazaka, Cologne, Germany) was used. It uses a specific UV-light camera to visualize the lesions of acne fluorescent an area of 8 x 10 mm. Three images were obtained at the same location in each region (forehead, cheek, and chin) and conducted by counting the number of orange-red fluorescent spots in each image using the device’s software. These points are porphyrin's endogenous metabolites of the bacteria P. acnes. Statistical analysis was used to average points scored between the three images (12).
Finally, at the end of the study, perception of efficacy was analyzed through a questionnaire that compared and analyzed the skin before and after treatment in terms of reducing oiliness, blackheads, acne and improvement in skin hydration and smoothness.
Statistical Analysis
The statistical analysis was performed according to the data distribution. For the normal distribution, the ANOVA test was applied. For the nonparametric, the Friedman test was performed using the GraphPad Prism 5 software.
Results and Discussion
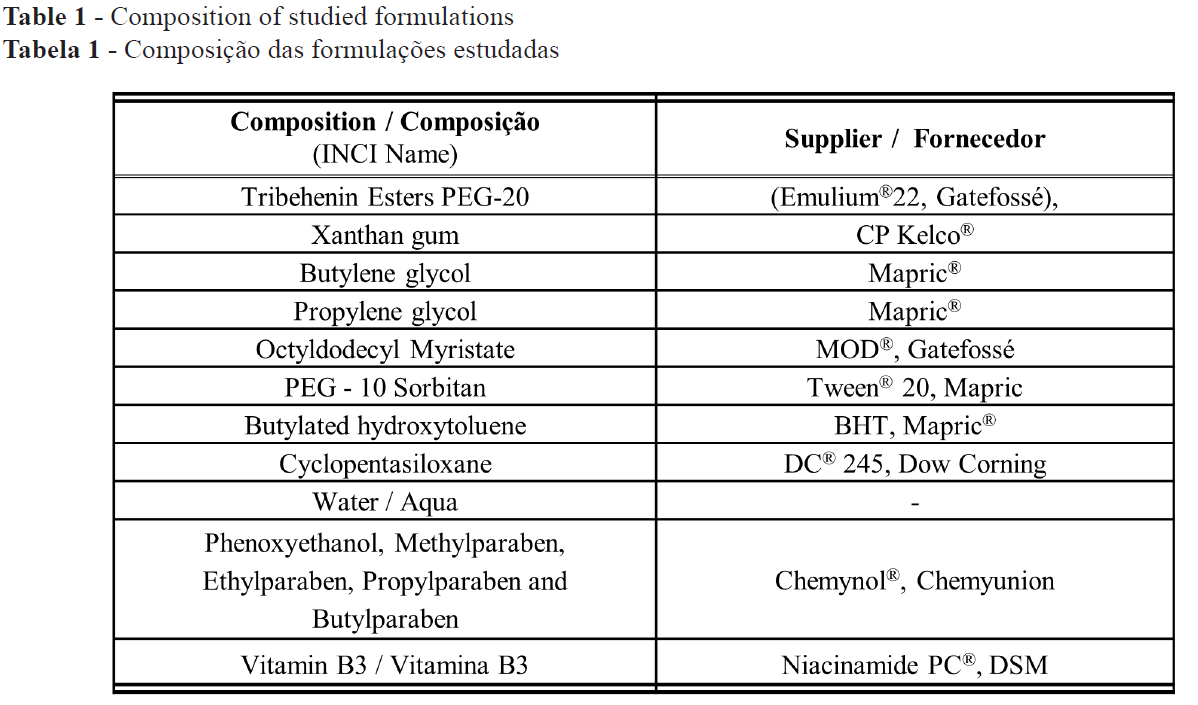
After statistical analysis of the parameters evaluated in the long-term efficacy study, it was observed that the formulation containing vitamin B3 showed a significant reduction of the amount of sebum after 28 days of application when compared to baseline values (Figure 1). This effect was not observed in the groups utilizing the vehicle.
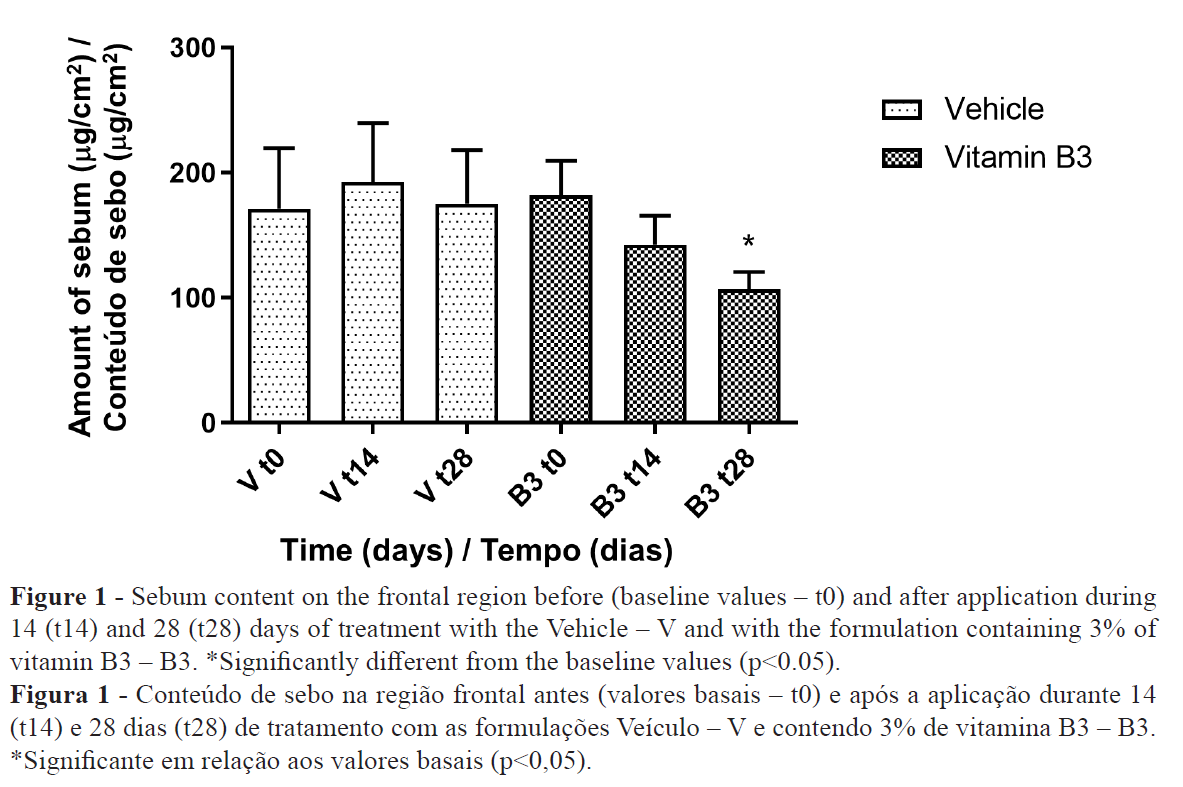
A decrease in the number of spots of porphyrin in the group that utilized the vitamin B3 formulation was also observed (Figure 2). The statistical analysis showed that the results were more pronounced in the frontal region of the face.
No significant alterations were observed in the Transepidermal Water Loss.
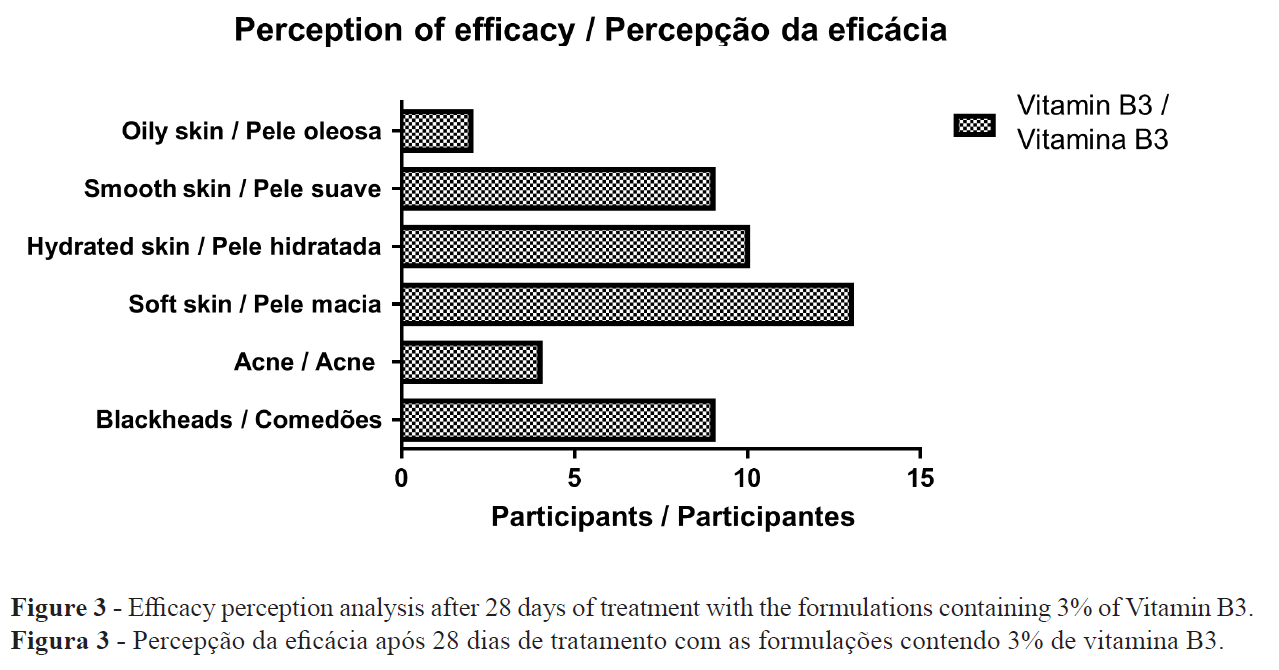
In the evaluation of the perception of effectiveness, it was possible to observe that the study participants perceived a reduction in the amount of sebum, blackheads and acne after treatment with the formulation containing vitamin B3. In addition, an improvement in skin hydration and smoothness was reported (Figure 3).
Topical nicotinamide has been shown to be useful in the treatment of acne vulgaris due to its sebo-suppressive, anti-inflammatory, and healing properties. The evaluation of the results showed that the topical application of formulations containing nicotinamide presented a significant reduction in the amount of sebum after and also a significant reduction in the porphyrin count after 28 days of application. This result can be explained by the reported anti-microbiological effect (13).
A previous study of our research group (12) showed that the presence and amount of porphyrins on the face are related to the secretion of sebum on the skin and a positive correlation among these features was found. In this context, the reduction of sebum along with the secretase of porphyrin count on the skin corroborates with the previous results of our research.
Alterations on the skin microbiome related to skin type have also been reported in the literature, where an increase in Cutibacterium acnes bacteria is observed in individuals with oily skin, as the bacteria consume this oil (14).
In this context, the present study showed that the use of 3% vitamin B3 presented a significant reduction in sebum content and porphyrins spots after 28 days of application. In addition, previous studies (15) reported that topical use of nicotinamide has been shown to improve fine lines and wrinkles, hyperpigmented spots, red blotchiness, and sallowness (yellowing), as well as skin elasticity.
An increase of the skin’s production of collagen and ceramides, stimulating keratinocyte differentiation, has also been reported, leading to an improvement of barrier function and skin appearance. It is important to highlight that the use of nicotinamide is comparable to topical antibiotic treatment with clindamycin gel (16), with the advantage that the side effects of nicotinamide are much safer than those of antibiotics substances and that it does not create antibiotic-resistant pathogens. This way, the use of vitamin B3 can be presented as an excellent alternative for the treatment of oily skin (17).
These effects have a great importance to the dermatological area as it contributes to the treatment of oily skin as a whole, without the use of any of the classical substances for these skin conditions which are usually more aggressive to skin health as a whole. Furthermore, the addition of substances that can improve the skin barrier function, keeping the maintenance of skin layers, is very important, as it does not cause damages while treating the excess of sebum.
At the same time, in the efficacy perception evaluation, the participants reported that their skin did not feel oily and a reduction in the presence of acne and blackheads was observed. Also, the participants also reported that they perceived their skin to be more hydrated, smooth and soft. The perceived efficacy is of great importance, as the observed effects by the participants support adherence to the treatment and consequently the efficacy with the application of the formulation (12).
In summary, after a statistical analysis of the obtained data, the formulation under study containing vitamin B3 showed a significant reduction of the amount of sebum when compared to baseline values. A reduction in the porphyrin count was also observed, with no alterations to the skin barrier. The applied questionnaires showed that the participants felt an improvement in the overall skin appearance and well-being while using the formulation under study.
In conclusion, the vitamin B3 was effective for application within formulations for oily skin care, reducing the amount of sebum and the porphyrin count while improving the appearance of skin and the participants well-being. Formulations containing vitamin B3 may be a excellent alternative for the treatment of oily skin, due to its complementary properties in improving skin conditions and women's quality of life.
Finally, the study has as its contribution the clinical, analytical evidence of the benefits of the topical use of vitamin B3 for the control of oily skin.
Author Contributions Statement
Study conceptualization: Patricia M.B.G. Maia Campos. Methodology: Patricia M.B.G. Maia Campos and Marcella G.A. Leite. Formal analysis: Patricia M.B.G. Maia Campos and Marcella G.A. Leite. Writing: Patricia M.B.G. Maia Campos and Marcella G.A. Leite. Writing – review and editing: Patricia M.B.G. Maia Campos.
Acknowledgements
The authors would like to express their thanks to Fapesp – Fundação de Amparo à Pesquisa do Estado de São Paulo and CNPq – Conselho Nacional de Desenvolvimento Científico e Tecnológico.
Conflict of Interests
Editors involved in this manuscripts’ authorship had no participation in the review or decision process. All authors have stated that there are no financial and/or personal relationships that could represent a potential conflict of interest.
References
1. Sakuma, T.H., Maibach, H.I. (2012) Oily Skin: An Overview. Skin pharmacology and physiology, 25(5), 227–235. https://doi.org/10.1159/000338978
2. Nouveau-Richard, S., Zhu, W., Li, Y. H., Zhang, Y. Z., Yang, F. Z., Yang, Z. L., Lian, S., Qian, B. Y., Ran, Y. P., Bouillon, C., Chen, H. D., & de Lacharrière, O. (2007). Oily skin: specific features in Chinese women. Skin research and technology : official journal of International Society for Bioengineering and the Skin (ISBS) [and] International Society for Digital Imaging of Skin (ISDIS) [and] International Society for Skin Imaging (ISSI), 13(1), 43–48. https://doi.org/10.1111/j.1600-0846.2006.00185.x
3. Wu, Y., Niu, Y., Zhong, S., Liu, H., Zhen, Y., Saint-Leger, D., Verschoore, M. (2013) A preliminary investigation of the impact of oily skin on quality of life and concordance of self-perceived skin oiliness and skin surface lipids (sebum). International Journal of Cosmetic Science, 35, 442–447. https://doi.org/10.1111/ics.12063
4. Wohlrab, J., Kreft, D. (2014) Niacinamide – Mechanisms of Action and Its Topical Use in Dermatology. Skin Pharmacology and Physiology,27, 311–315. https://doi.org/10.1159/000359974
5. Khodaeiani, E., Fouladi, R.F., Amirnia, M., Majid, S., Karimi, E.R. (2013) Topical 4% nicotinamide vs. 1% clindamycin in moderate inflammatory acne vulgaris. International Journal of Dermatology, 2013, 52: 999–1004.https://doi.org/10.1111/ijd.12002
6. Shalita, A.R., Smith, J.G., Parish, L.C. (1995) Topical nicotinamide compared with clindamycin gel in the treatment of inflammatory acne vulgaris. International Journal of Dermatology, 34, 434–437. https://doi.org/10.1111/j.1365-4362.1995.tb04449.x
7. Chen, A.C., Damian, D.L. (2014) Nicotinamide and the skin. Australasian Journal of Dermatology, 55, 169–175. https://doi.org/10.1111/ajd.12163
8. Andrade, J.P., Wagemaker, T.A.L., Mercurio, D.G., Maia Campos, P.M.B.G. (2018) Benefits of a dermocosmetic formulation with vitamins B3 and a B6 derivative combined with zinc-PCA for mild inflammatory acne and acne-prone skin. Biomedical and Biopharmaceutical Research, 15(2), 214-223. doi: 10.19277/bbr.15.2.188
9. Maia Campos, P.M.B.G., Melo, M.O., Calixto, L.S., Fossa, M.M. (2015) An Oral Supplementation Based on Hydrolyzed Collagen and Vitamins Improves Skin Elasticity and Dermis Echogenicity: A Clinical Placebo-Controlled Study. Clinical Pharmacology Biopharmaceutic, 4, 142. doi:10.4172/2167-065X.1000142
10. Rogiers, V., EEMCO Group. (2001) EEMCO guidance for the assessment of transepidermal water loss in cosmetic sciences. Skin Pharmacology and Physiology, 14(2), 117- 128. https://doi.org/10.1159/000056341
11. Crowther, J.M. (2015) Method for quantification of oils and sebum levels on skin using the Sebumeter®. International Journal of Cosmetic Science,38, 210–216. https://doi.org/10.1111/ics.12258
12. Gabarra Almeida Leite, M., & Maia Campos, P. (2020). Correlations between sebaceous glands activity and porphyrins in the oily skin and hair and immediate effects of dermocosmetic formulations. Journal of cosmetic dermatology, 19(11), 3100–3106. https://doi.org/10.1111/jocd.13370
13. Rolfe H. M. (2014). A review of nicotinamide: treatment of skin diseases and potential side effects. Journal of cosmetic dermatology, 13(4), 324–328. https://doi.org/10.1111/jocd.12119
14, Zheng, Y., Liang, H., Zhou, M., Song, L., He, C. (2021) Skin bacterial structure of young females in China: The relationship between skin bacterial structure and facial skin types. Experimental Dermatology, 30(10),1366-1374. https://doi.org/10.1111/exd.14105
15. Tran, D., Townley, J.P., Barnes, T.M., Greive, K.A. (2015) An antiaging skin care system containing alpha hydroxy acids and vitamins improves the biomechanical parameters of facial skin. Clinical, Cosmetic and Investigational Dermatology, 8, 9–17. https://doi.org/10.2147/CCID.S75439
16, Khodaeiani, E., Fouladi, R.F., Amirnia, M., Saeidi, M., Karimi, E.R. (2013) Topical 4% nicotinamide vs. 1% clindamycin in moderate inflammatory acne vulgaris. International Journal of Dermatology, 52, 999–100. https://doi.org/10.1111/ijd.12002
17. Wohlrab, J., Kreft, D. (2014) Niacinamide - Mechanisms of Action and Its Topical Use in Dermatology. Skin Pharmacology and Physiology, 27, 311-315. https://doi.org/10.1159/000359974
18. Bonilha, G.C., Costa, G.M.D. Maia Campos, P.M.B.G. (2020) Rheological, texture, and sensory analyses and in vivo clinical efficacy of cosmetic formulations containing ascorbyl tetraisopalmitate. Biomedical Biopharmaceutical Research, 17(1), 1-12. doi: 10.19277/bbr.17.1.228
